Wall Street Wants the Best Patents, Not the Best Drugs
(Bloomberg Opinion) -- It’s probably unfair to start a column about the new drugs that are curing Hepatitis C by referencing Jonas Salk and the discovery of the polio vaccine, but I’m going to do it anyway. It’s worth remembering what the world used to look like as we contemplate what it looks like now.
In the late 1940s and early 1950s, there was nothing that scared American parents more than polio, a disease that (a) caused partial paralysis, (b) was easily transmitted, and (c) primarily affected children. Salk, who had worked on flu vaccines during World War II, joined the University of Pittsburgh in 1947 and soon began working on a possible polio vaccine. In 1953, he announced — on a national radio broadcast, no less — that he had developed a vaccine that prevented the disease; two years later, once its efficacy had been proved, the country undertook a national inoculation program, paid for by the federal government.
Did his discovery of the polio vaccine make Salk rich? It did not. When the CBS journalist Edward R. Murrow had Salk on his interview show, “See It Now,” he asked the scientist, “Who owns the patent on this vaccine?”
“The people, I would say,” Salk replied, after a moment’s hesitation. “There is no patent. Could you patent the sun?”
In 2014, Gilead Sciences Inc. introduced Sovaldi, the first drug that cured Hepatitis C, a liver disease that may affect as many as 150 million people worldwide, and that can be life-threatening if untreated. As is always the case in the modern age, Sovaldi was surrounded by a thicket of patents, some of which wouldn’t expire until 2034.
The company knew, however, that AbbVie Inc. was less than a year away from coming out with its own Hep C drug, and thus its monopoly was likely to be short-lived. Under pressure to capitalize on its invention during a short window, the company priced Sovaldi at $84,000 for a 90-day course of treatment. Less than a year later, the Food and Drug Administration approved a second Gilead drug, Harvoni, which in some cases could cure Hep C patients in 60 days instead of 90. Harvoni cost $94,000.
The reaction was predictable. Hep C sufferers expressed outrage at the price. Insurers — including Medicare — balked at paying so high a price; even after rebates the price was still over $50,000. Some of them limited the drug to only to the sickest patients.
The Senate Finance Committee conducted an investigation and concluded that Gilead’s pricing strategy was “a calculated scheme” aimed at “maximizing revenue.” Gilead, the committee wrote, “knew these prices would put treatment out of reach of millions and cause extraordinary problems for Medicare and Medicaid, but the company still went ahead.”
But because the drugs worked so well, most insurers swallowed hard and paid for them. Even after AbbVie came out with its first Hep C drug, the Gilead drugs remained dominant, largely because the AbbVie products didn’t work as well. Between 2014 and 2017, Gilead’s generated $50 billion from its Hep C “franchise,” curing hundreds of thousands of patients while leaving rivals in the dust.
I first starting paying attention to Gilead’s Hep C drugs in the spring of 2016, when I noticed that its stock had begun to drop precipitously. I couldn’t understand why Wall Street was down on a company with a near-monopoly on a drug that actually cured a disease rather than simply treated symptoms, as most drugs do. Isn’t a cure the holy grail of drug discovery?
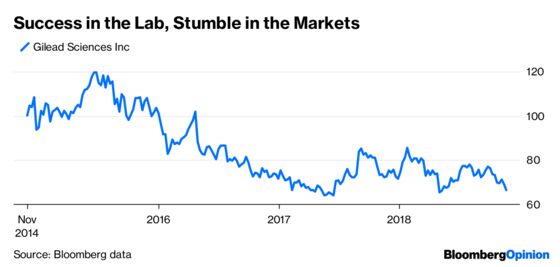
For patients and doctors, it certainly is. But it turns out that Wall Street much prefers a drug like AbbVie’s Humira, which treats — but doesn’t cure — rheumatoid arthritis, Crohn’s disease and a variety of other illnesses. Patients who need it use it forever. With an estimated $38,000-a-year price tag (after rebates), and patents that won’t expire for years, it is the biggest selling drug in the world.
Gilead, meanwhile, began to see its patient population dwindle as its Hep C drugs worked their magic. There are only 3.5 million chronic Hep C sufferers in the U.S., but many of them are in prison or on Medicare or Medicaid, which had rules that made using the drug difficult. By some estimates, as many as 85 percent of the people with chronic Hep C lacked access to treatment. In addition, while Gilead allowed generic manufacturers to make Hep C competitors in many poorer countries, it declined to do so in such countries as Argentina, Brazil, China and most of Western Europe.
Enter a group called the Initiative for Medicines, Access and Knowledge, or I-MAK for short. I-MAK is funded in part by John Arnold, a billionaire energy trader who once worked at Enron. Its mission is to challenge key pharma patents in an effort to enable generic competition and bring down the price of important, life-saving drugs. Tahir Amin, one of I-MAK’s co-founders, told me that his group had had its eye on Sovaldi as long ago as 2012, even before the drug received approval by the Food and Drug Administration. I-MAK’s position is that the basic compound at the heart of Gilead’s drugs, sofosbuvir, is simply not novel enough to merit 20 years patent protection.
In the U.S. that argument hasn’t gotten very far; despite numerous challenges, Gilead’s patents have been sustained by the U.S. patent office. But elsewhere I-MAK had considerable success.
In 2015, in the face of an I-MAK patent challenge in India, Gilead decided to allow generics to copy its Hep C drugs. Ukraine rejected a key Gilead patent. Argentina turned thumbs down on Gilead’s patents. In May, a court in Brazil rejected an important Gilead patent. And most recently, in August, I-MAK's challenge succeeded in getting Gilead to withdraw key patent claims to sofosbuvir in China, where 8.9 million people have chronic Hep C. All of these victories have increased the likelihood that low-priced generics will replace Gilead’s Hep C drugs in those nations.
I told Amin that it struck me as a little unfair that a company that actually cured a disease was getting hammered like this while AbbVie’s gaming of the patent system to maintain its Humira monopoly has gone largely unchallenged. At the very least, I had a hard time seeing why any pharma company would want to work to find cures. All the incentives seemed to skew in the direction of drugs that treated symptoms – and would have to be used for years.
“This is certainly a question we’ve thought about,” Amin responded. “But drugs aren’t annuities. The problem is that the patent system has allowed companies to keep extracting payments where they either don't deserve a patent or where they are overpatenting well beyond the intended 20-year term.” He added: “When you have a drug with a list price of $84,000, you can’t just stand back and let people die because they can’t afford it.”
Amin told me that the real problem is that the incentive structure is all wrong under the current patent system: There needs to be a way to give drug companies an incentive to invest in potential cures and get a return that doesn’t depend on a 20-year patent.” The Gilead Hep C story is certainly a case in point, from the outrageous early prices, to the Senate investigation, to Wall Street’s disapproval, to the strenuous effort to remove the patent protection abroad.
The truth is, though, as pharmaceutical development has become ever more oriented to Wall Street, protection from generics is the only bankable asset.
Could you patent the sun? No need. The universe of Big Pharma now orbits around patents themselves.
To contact the editor responsible for this story: Philip Gray at philipgray@bloomberg.net
This column does not necessarily reflect the opinion of the editorial board or Bloomberg LP and its owners.
Joe Nocera is a Bloomberg Opinion columnist covering business. He has written business columns for Esquire, GQ and the New York Times, and is the former editorial director of Fortune. He is co-author of “Indentured: The Inside Story of the Rebellion Against the NCAA.”
©2018 Bloomberg L.P.