Patient Hurt by Do-It-Yourself Artificial Pancreas Prompts FDA Warning
Patient Hurt by Do-It-Yourself Artificial Pancreas Prompts FDA Warning
(Bloomberg) -- The U.S. Food and Drug Administration warned diabetics against building their own artificial pancreas system to help control blood sugar levels after a patient using one suffered an accidental insulin overdose.
A large community of diabetics has been using hacked-together, do-it-yourself systems to control their disease. The systems connect glucose monitors to insulin pumps using computer algorithms. They work around the clock, testing blood sugar and infusing insulin. Once the system is set up, they’re meant to require little effort by the patient.
While the individual components that make up a DIY system are generally approved by the FDA, they haven’t been tested or cleared for use together, the agency said in a statement. The accidental insulin overdose didn’t kill the patient, an FDA spokeswoman said.
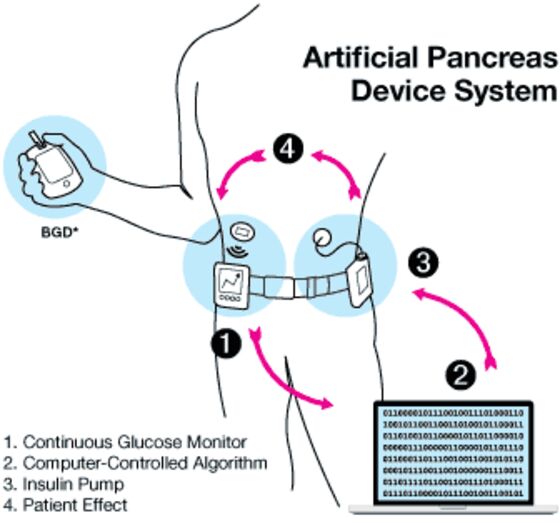
“Patient use of unauthorized diabetes-management devices, alone or along with other devices, could result in inaccurate glucose level readings or unsafe insulin dosing,” the FDA said. “These inaccuracies may lead to injuries requiring medical intervention, such as severe low blood sugar, coma, diabetic ketoacidosis -- the buildup of acids in blood -- and death.”
The FDA warning is an example of the tension between patients who want to take more of their health care into their own hands and increasingly have the technological tools and know-how to do so, and regulators charged with safeguarding public health.
Still, it’s the first warning from the agency involving the do-it-yourself systems, which have been gaining traction for the past several years and currently have thousands of users. Some diabetics have trouble controlling their blood sugar and insulin levels even when using only FDA approved devices, and in some cases require medical attention or hospitalization.
Patient Injury
The FDA received a report about a patient using one of the unauthorized systems who got too much insulin after getting repeated, incorrect readings of high blood sugar levels, causing an overdose and requiring medical intervention.
“These devices were not designed to be used together and were combined in a way that had not been thoroughly tested for compatibility,” the agency said. It’s not clear if the problem stemmed from inaccurate glucose values or a software malfunction.
The systems are informally known as an artificial pancreas, since the pancreas is the organ that produces insulin needed to convert blood sugar into energy. Patients with type 1 diabetes, the most severe form, don’t produce any insulin naturally. Instead, they have to regularly inject it to avoid glucose buildup that eventually damages the heart and blood vessels, and causes complications including blindness, kidney failure and amputations. Too much insulin, however, can cause an overdose -- leading to a coma or death.
Medtronic Plc makes an approved version of the technology, called the 670G. While closely regulating insulin levels, it doesn’t allow as much flexibility as the systems that individuals make themselves.
The FDA said that some companies had been marketing unauthorized software that shows patients a glucose levels pulled from raw data. The agency didn’t identify the companies, but said it could result in inaccurate values being shown to the patient.
To contact the reporter on this story: Michelle Fay Cortez in Minneapolis at mcortez@bloomberg.net
To contact the editors responsible for this story: Drew Armstrong at darmstrong17@bloomberg.net, Rick Schine, Michael Hytha
©2019 Bloomberg L.P.