Danish Meme Stock Plunges as Failed FDA Bid Triggers Panic
Danish Meme Stock Orphazyme Cuts Outlook After Failing FDA Test
(Bloomberg) -- Orphazyme A/S, a small Danish biotech firm that morphed into a meme stock last week, was dumped by investors on Friday after it failed to get a clear stamp of approval from U.S. authorities for its only viable treatment.
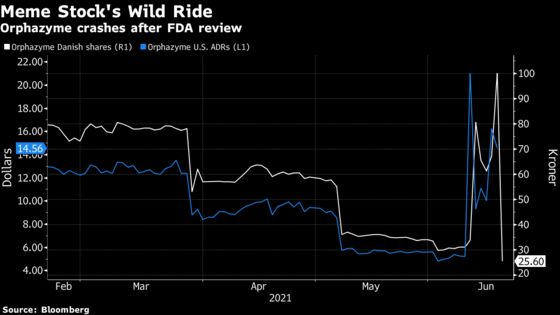
The company’s American depositary shares lost almost half their value once trading started in the U.S., matching a selloff in Copenhagen earlier in the day. The move brings its market cap to about $300 million, compared with $560 million less than 24 hours ago.
This is “game-over” for all those who’d speculated that Orphazyme might soar on sudden regulatory approval, said Per Hansen, an investment economist at retail broker Nordnet. It’s been a period of “high-stakes gambling” and even if U.S. authorities had given their approval, “it wouldn’t at all justify” the recent gains in the stock, he said.
In a statement on Friday morning, the Copenhagen-based company said it is “disheartened” by the Food and Drug Administration’s decision to seek further information before clearing its treatment for Niemann-Pick disease type C.
“We remain committed to working with the regulators” to win approval for arimoclomol, Chief Executive Officer Christophe Bourdon said. Orphazyme is still hoping for European regulatory clearance, which may come in the fourth quarter, he said.
But the news will have a “significant” impact on Orphazyme’s finances, the CEO acknowledged. The company says it may see losses seven times as deep as originally feared, and it only expects to have about 50 million kroner ($8 million) in cash by the end of 2021, one seventh its previous guidance.
“In the short-term, we will need to reduce our costs substantially and freeze all company efforts” that aren’t tied to getting its one treatment approved, the company said.
Just one week ago, Orphazyme was embraced by investors on social media platforms such as Reddit, helping drive its shares almost 1,400% higher at one point during U.S. trading hours. Since then, the stock has tested the extremes of volatility, and the company itself has warned investors against being sucked into the frenzy.
Denmark’s Financial Supervisory Authority says it won’t comment on Orphazyme specifically, but warned that it will step in should there be any indication of deliberate “price manipulation.” The watchdog “always reacts” to specific concerns, Anders Balling, a director at the FSA in Copenhagen, said by email earlier this week.
“The situation is hopeless,” Hansen at Nordnet said. The company has already failed to win approval on two previous occasions, “and now the FDA is providing the coup de grace.”
To view the source of this information click here
Story Link: Orphazyme Says FDA Needs Additional Data for Arimoclomol
©2021 Bloomberg L.P.