Solid Bio’s Plunge Sends a Warning on Gene Therapies
Solid Bio’s Plunge Sends a Warning on Gene Therapies
(Bloomberg Opinion) -- It’s easy to get excited about gene therapies, a group of new medicines that have the potential to cure diseases in one treatment. The saga of Solid Biosciences Inc. is a reminder that tweaking genes isn’t a trivial task. These therapies are full of promise, but they’re not entirely there yet.
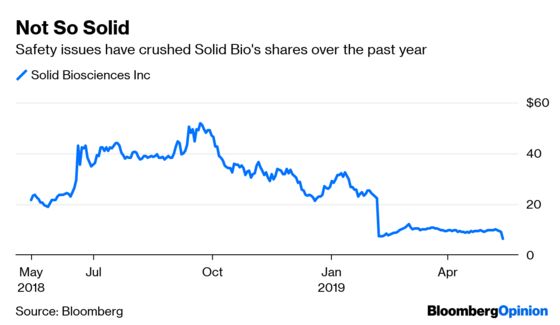
Solid Bio has been working on a gene therapy for a muscle-wasting disease. It went public last year and saw its market value climb to almost $2 billion amid a steady rise in its shares, only to plunge in recent months as its once-hyped efforts have been stymied by side effects and weak data. Then late Monday, it disclosed more potentially worrisome safety data after upping the dose, triggering a plunge in its stock of as much as 40 percent on Tuesday. It’s now worth around $200 million.
Gene-therapy fever has led to a wave of expensive buyouts and lucrative IPOs. The excitement around these treatments has led to a great profusion of gene-therapy efforts in a variety of diseases, some of which are likely to end up like Solid Bio’s. But it’s still very early days.
While safety issues tripped up Solid Bio, they aren’t the only risks. There are also concerns about whether these medicines are durable cures. Biomarin Pharmaceutical Inc.’s gene therapy for treating hemophilia has blockbuster potential, for instance, but its latest trial results showed that levels of the clotting factor it helps patients produce declined over time. Investors are watching closely for any further erosion, and billions of dollars in value and sales potential may swing on the result.
These durability fears raise another issue related to gene therapies: their cost. They are designed as one-time treatments for usually rare diseases, so prices are high. Spark Therapeutics Inc.’s eye-disease gene therapy Luxturna costs $425,000 per eye and has struggled to make commercial headway, though that didn’t stop Roche Holding AG from acquiring the company for $4.8 billion.
Some gene therapies – like Biomarin’s hemophlia drug – could replace expensive treatments that must be taken for a lifetime, so there’s justification for a high price. That case is diminished if insurers doubt their durability, especially if drugmakers remain reluctant to offer deep discounts when their medicines don’t work.
The massive potential of gene therapy means that money is easy and optimism is high right now. But the youth of this field and the dearth of actual commercial success means that it needs to be treated with caution for some time to come.
To contact the editor responsible for this story: Beth Williams at bewilliams@bloomberg.net
This column does not necessarily reflect the opinion of the editorial board or Bloomberg LP and its owners.
Max Nisen is a Bloomberg Opinion columnist covering biotech, pharma and health care. He previously wrote about management and corporate strategy for Quartz and Business Insider.
©2019 Bloomberg L.P.