Sinopharm Jumps Most in 5 Years on Coronavirus Vaccine Data
Sinopharm Jumps Most in 5 Years on Coronavirus Vaccine Results
(Bloomberg) -- Sinopharm Group Co. surged by the most since April 2015 on Wednesday to help lead a rally among Chinese health-care names, as the trial results for a Covid-19 vaccine candidate rekindled investors’ enthusiasm for the sector.
The vaccine candidate, developed by Wuhan Institute of Biological Products Co., has shown no serious adverse reactions during phase I and II clinical trials, according to a Weibo posting Tuesday by China National Biotec Group, a subsidiary of the company’s parent. That sent Sinopharm shares up by as much as 15% in Hong Kong before finishing 9.2% higher.
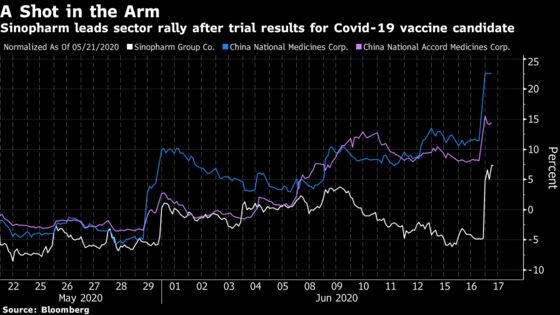
China National Medicines Corp., a unit of Sinopharm, jumped the 10% limit in Shanghai while another subsidiary, China National Accord Medicines Corp., also rose the daily maximum in Shenzhen. Six of the top 10 gainers in the Asia Pacific region were Chinese pharmaceutical or biotechnology firms, including Sinopharm and the two units. Meanwhile, the health-care segment in the MSCI China Index rose 2.1%, hitting a fresh two-year high.
Using a killed version of the coronavirus, the vaccine developed by Sinopharm is among five Chinese experimental shots that have reached the crucial final stage of human testing before they can be approved for public use. Data collected from early phases of human testing in three of the five vaccines have suggested they are safe and able to elicit immune response to the virus in studies that enrolled hundreds of people.
As the world races to develop a vaccine against the coronavirus, Beijing has mobilized its health authorities, drug regulators and research institutes to work around the clock with local firms. China has promised to share any successful vaccine globally.
©2020 Bloomberg L.P.
With assistance from Bloomberg