Is a Blood Test for Multiple Cancers Hope or Hype?
(Bloomberg Opinion) -- More than half a million Americans die of cancer each year, matching the toll of Covid-19 to date on an annual basis and making it one of the nation's biggest killers. But now there’s hope that a simple blood test could change that by detecting tumors earlier when they’re easier to treat.
Grail Inc., a biotechnology startup, launched a blood test called Galleri in the U.S. in June and published data validating its ability to detect otherwise hard-to-find tumors. While blood tests already exist for individual cancers, Grail's caught 13 types of the disease in a real-world experiment. If these tests prove out it could slash cancer's burden so unsurprisingly, the news got a lot of attention. But is it more hype than hope? There’s promise, for sure. It's essential, though, to acknowledge some realities of cancer screening that may hold these tests back.
It seems obvious that we ought to search for and catch as many cancers as possible, right? Well, not exactly. As counterintuitive as this may sound, that strategy can be wasteful and even harmful on a population level. The history of cancer testing includes life-saving successes — such as pap smears for cervical cancer — but also costly misses like prostate and thyroid screening, which don't always have strong benefits. The result is that we routinely look for only a few cancers.
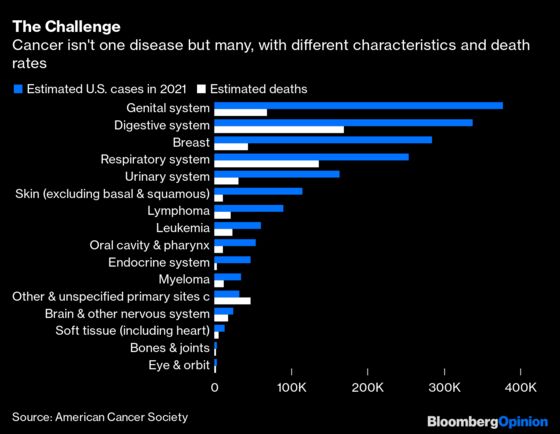
What makes cancer screening so tricky? Even accurate tests don't perform as well when looking for comparatively rare diseases like cancer. That leads to false positives, resulting in costly follow-up and an emotional toll. That's why the few recommended screenings look for relatively common cancers in higher-risk populations. Additionally, as odd as it sounds, some slow-growing tumors aren’t dangerous, so testing that identifies them leads to expensive overtreatment. There’s also a phenomenon known as "lead-time bias,” where screening can lead to an earlier diagnosis but not a better outcome. It's a tricky concept that requires a bit of a thought experiment to understand. Imagine that person A and person B developed similar cancers in 2015. Person A gets screened in 2016, and their cancer is treated. Person B doesn't find out they have the disease until 2021. If person A relapses and both die of cancer in 2022, screening may be credited with adding six years when it didn't truly do much.
All of this creates the need for a careful evaluation and a tradeoff when it comes to screening, especially if casting a wide net identifies more cancers but at the likely cost of more incorrect or unproductive positive results. But the multi-cancer blood tests like Grail's and others in development from companies such as Exact Sciences Corp. are especially exciting because they could change the testing math enough to make broader screening possible. Gene sequencing giant Illumina Inc. certainly sees the potential: It agreed to buy Grail for $8 billion if antitrust regulators let it.
Galleri, which identifies bits of tumor DNA in the blood, demonstrated a very low false-positive rate in validation studies, especially in comparison with many existing screening methods. Along with the fact that it’s searching for multiple cancer types instead of just one, that should help these tests be used widely without too many wrong answers. Quick detection, in turn, could make cancer cases both more curable and potentially a lot cheaper. Doctors can often treat earlier cancer with surgery and radiation. In later stages, the only resort is often costly medicines that don't always work very well. Early evidence also suggests that Grail's test may be more likely to catch concerning tumors.
The big barrier between these exciting features and wide embrace of the technology is proof, and the fact that the medical profession has been burned before by the promise of early detection. There needs to be more well-controlled evidence and expert evaluation to validate these tests before anyone can confidently declare that this time is different.
The next few years could provide valuable answers and speed adoption. Grail is running studies in different populations, and others will follow. The U.K.’s National Health Service is conducting an ambitious 140,000 person randomized trial of Galleri that should be particularly helpful in understanding the real-world performance and use cases of the test. The Brits have the right idea: A large demonstration is an excellent way to get real use out of the test while generating valuable data.
The U.S. government should join the effort, especially given that it foots the bill for a significant portion of cancer costs via Medicare. Funding further large trials could help compare tests, evaluate use in more groups and help answer the ultimate question of whether blood screening saves lives.
None of this will be easy or cheap, and the studies won’t deliver firm answers right away. But the potential benefit is big enough to make an effort worthwhile.
This column does not necessarily reflect the opinion of the editorial board or Bloomberg LP and its owners.
Max Nisen is a Bloomberg Opinion columnist covering biotech, pharma and health care. He previously wrote about management and corporate strategy for Quartz and Business Insider.
©2021 Bloomberg L.P.