(Bloomberg Opinion) -- Thierry Breton, the European Union commissioner in charge of fixing the bloc’s mass vaccination campaign, likes to start his day with a call to AstraZeneca Plc boss Pascal Soriot to keep tabs on the supply of Covid-19 vaccines critical to immunizing the bloc’s population and reopening its economy by the summer.
The conversation’s been tense for a while, but surely more so this week. Countries across Europe including France, Germany and Italy have suspended vaccination with AstraZeneca’s Covid-19 shot following reports of serious blood clotting. The European Medicines Agency still says the benefits of being vaccinated outweigh the risks. It’s looking into the reports. The nations hitting pause say they’re doing it for purely “precautionary” reasons — to be safe rather than sorry.
This is a complex issue, with few easy answers. The EU is facing a new wave of Covid-19 infections, and the AstraZeneca vaccine is a critical weapon against the coronavirus. Any interruption to the rollout is an immediate setback. The need to protect citizens from danger also means health authorities are “stuck between a rock and a hard place,” as Bloomberg Intelligence’s Sam Fazeli puts it.
If this uncertain situation isn’t resolved quickly, the risk is that the precautionary principle used to explain such decision-making will do more harm than good, especially at a time when more lockdowns loom.
Investigating reported side-effects and severe events is vital in a vaccination campaign, but there’s a real question as to whether slamming on the brakes in this instance unnecessarily downplays the data we already have. As of March 10, 30 cases of blood-clot events had been reported among close to 5 million Europeans given the AstraZeneca shot, according to the Amsterdam-based EMA, or 0.0006%.
AstraZeneca on Monday said there was no evidence of increased blood-clot risk among those vaccinated in the U.K. and EU, and that in clinical trials the number of events was actually lower in those vaccinated compared with the placebo group.
There’s a fine line between precaution and what the University of Copenhagen’s Ezio Di Nucci, an expert on medical ethics, warns is a “zero-risk” worldview — one that might bring second-guessing at every turn in the global vaccination campaign.
One could potentially argue that a belt-and-braces approach means going above and beyond to preserve public trust, especially in countries with traditionally high vaccine hesitancy like France. Any error might prove fateful to the public take-up of Covid-19 vaccines.
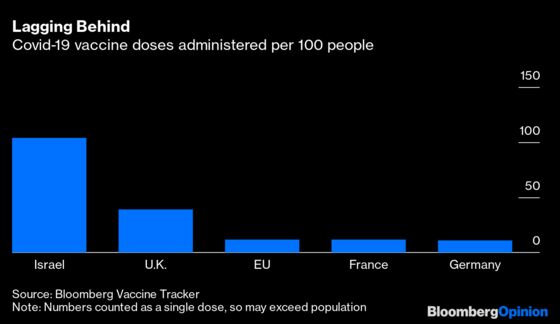
Yet their caution runs directly counter to explicit warnings by public-health regulators like the EMA and the World Health Organization of the risk of delaying inoculations, bringing more cases and deaths. And politicians’ shifting positions on the safety and efficacy of vaccines like AstraZeneca’s can even erode the confidence they’re working so hard to win over.
While this precautionary urge comes “from the right place,” according to Maxwell Smith, assistant professor at Western University in Canada, the threshold to stop Covid vaccination programs could be set higher. A more proportionate approach might weigh the epidemiological and economic impact: Insurer Allianz SE has estimated the cost of a five-week delay to the EU’s economic reopening at 90 billion euros ($107 billion).
Europeans would do well to weigh the knock-on impact for the rest of the world too, so soon after reaffirming the EU’s commitment to sharing doses with developing countries via the Covax multinational initiative and so soon after Italy blocked exports of AstraZeneca doses to Australia to satisfy domestic demand.
There are lessons here beyond vaccines. The plight of European politicians seems to underscore what Harvard’s Cass Sunstein once called the “paralyzing” nature of the precautionary principle, which can be invoked as both an obstacle to action as well as inaction. Or as one French public-health expert put it bluntly, “A precautionary principle is something you can apply when you don’t have a disease devastating the planet.”
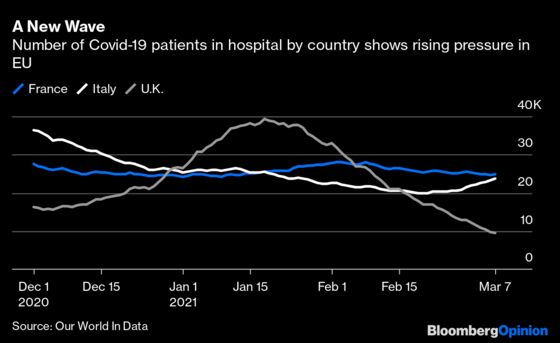
Take French President Emmanuel Macron’s foot-dragging over whether to impose a new lockdown: What he and his advisers consider as victories — another day or week without stay-at-home curbs — may actually be prolonging the pain and delaying the remedy. If U.K. Prime Minister Boris Johnson’s speed-first vaccination drive is explicitly modeled on the Steven Soderbergh movie “Contagion,” Macron’s could be equated to Ingmar Bergman’s “The Seventh Seal,” in which a knight plays chess with Death as a plague rages.
A middle ground between throwing caution to the wind and trying to avoid every potential danger is possible. Leaders need to stop ignoring the pandemic forest for the trees. That means putting less focus on low-probability risks and taking a more proportionate overall view of the trade-offs involved in delayed or hesitant decision-making.
Better communication and data can help. Ruth Faden, who founded the John Hopkins Berman Institute of Bioethics, says collecting good-quality data on drug safety and side effects around the world will be one way of improving decision-making around vaccination campaigns. European leaders have much of this in hand, and they should employ them to their advantage now.
Hopefully, regulators will draw a line under this issue in the coming hours and days. But with billions of people around the world due to be vaccinated, this has to be handled better next time — because there will be a next time. Otherwise the next conversation between Breton and Soriot will be a silent one.
This column does not necessarily reflect the opinion of the editorial board or Bloomberg LP and its owners.
Lionel Laurent is a Bloomberg Opinion columnist covering the European Union and France. He worked previously at Reuters and Forbes.
©2021 Bloomberg L.P.