Pfizer Ramps Up Production; Moderna Effectiveness: Virus Update
Track the global Covid-19 pandemic and the containment efforts here.

(Bloomberg) -- The U.S. expects to have enough vaccine to meet demand by May despite a planned pause in the use of Johnson & Johnson’s shot pending a review of rare blood clots. J&J said it’s delaying its rollout in Europe.
German Chancellor Angela Merkel’s cabinet approved a law setting nationwide rules on virus restrictions after some regions failed to impose curbs despite rising cases. Italy is in talks to set up vaccine production hubs as the government seeks to build domestic capacity. Meanwhile, the U.K. hit a goal to offer a shot to all over 50s three days early.
Key Developments:
- Global Tracker: Cases pass 137 million; deaths 2.95 million
- Vaccine Tracker: More than 814 million shots given worldwide
- Blood Clots, Anaphylaxis and Other Vaccine Fears: QuickTake
- Why the mutated coronavirus variants are so worrisome
- What we know about the impact of Covid-19 on children
- Which vaccine is best, and other questions answered (Video)
Subscribe to a daily update on the virus from Bloomberg’s Prognosis team here. Click CVID on the terminal for global data on cases and deaths.
Walmart Halts Administration of J&J Vaccine (5:36 p.m. NY)
Walmart Inc. said it stopped administering the Johnson & Johnson vaccine after the U.S. Centers for Disease Control and Prevention and Food and Drug Administration said they would pause use of the shot after six women who received it developed a rare and severe form of blood clotting.
A Walmart spokesperson said in an email that the company will offer substitutes, when possible, and will cancel appointments if an alternative isn’t available.
Pfizer Ramps Up Production, CEO Says (4:47 p.m. NY)
Pfizer Inc. Chief Executive Albert Bourla said on Twitter that the company ramped up production and will be able to deliver 10% more doses to the U.S. by the end of next month than had been previously agreed on.
Moderna Shot 90% Effective After Six Months (4:35 p.m. NY)
Moderna Inc.’s vaccine remained more than 90% effective after six months, according to a new analysis of data from the company’s final-stage trial.
Beginning two weeks after the second dose, the shot was more than 90% effective overall, and more than 95% effective at preventing severe cases, according to a statement. The company didn’t release further details and said the follow-up results were preliminary as the study is continuing.
WHO Urges Pause in Sale of Live Animals (3:20 p.m. NY)
The World Health Organization on Tuesday urged countries to suspend the sale of wild animals at so-called wet markets as an emergency measure, saying 70% of all emerging infectious diseases in humans have wildlife origins. The first outbreak of Covid-19 was linked to a wet market in Wuhan, China.
“Although the specific mechanism of SARS-CoV-2 emergence has not been definitively identified, at some point or over time, interactions may have occurred that allowed for cross- and perhaps multiple-species pathogen transmission,” the United Nations agency said in a report.
The WHO acknowledged the central role traditional markets play in providing food and livelihoods for large populations. However, it said that “banning the sale of the animals can protect people’s health — both those working there and those shopping there.”
Biden Says There Are Enough Doses (2:50 p.m. NY)
Dutch Lockdown Measures Extended (1:45 p.m. NY)
Lockdown measures in the Netherlands will be prolonged until at least April 28 as the number of new Covid-19 cases and hospital admissions continue to strain the country’s health system, Prime Minister Mark Rutte said at a press briefing in The Hague on Tuesday evening. If fewer patients are admitted, Rutte’s government announced a plan to reopen Dutch society, with first steps including reopening outdoor seating areas and end to a much-debated night-time curfew.
France Suspends Flights to Brazil (1:25 p.m. NY)
France suspended all flights to and from Brazil until further notice as concern grows over the spread of the Brazilian variant of the virus.
France, which is under a national lockdown, is struggling to contain the pressure on its health system, with intensive-care unit occupancy remaining on a steady increase in the past few weeks, reaching 117.7% on Tuesday. France recorded 39,113 new infections and 345 new deaths in the past 24 hours.
NYC Rescheduling J&J Appointments (10:40 a.m. NY)
New York Mayor Bill de Blasio said the city would pause all Johnson & Johnson vaccinations on Tuesday following guidance from U.S. health officials.
The city will reschedule upcoming J&J appointments for residents, who will instead receive vaccines made by Pfizer Inc.-BioNTech SE and Moderna Inc., de Blasio said in a Tuesday briefing. De Blasio, who received the J&J shot himself, said the city has given 234,000 doses of the vaccine and hasn’t seen any reports of blood clots. Overall, New York has delivered more than 5 million doses as it seeks to fully vaccinate 5 million residents by June.
De Blasio made an appeal to the federal government to reallocate shots from regions with over-capacity. “We have endless need, endless demand,” he said.
J&J to Delay Rollout in Europe (9:22 a.m. NY)
Johnson & Johnson decided to delay the rollout of its Covid-19 vaccine in Europe. The company is reviewing blood clot cases with European health authorities.
Earlier, U.S. health officials recommended a pause in the use of the vaccine. Six women suffered a type of brain blood clot similar to that reported as a rare side effect to the AstraZeneca Plc vaccine. About 6.8 million people in the U.S. have received the J&J shot.
“This announcement will not have a significant impact on our vaccination plan -- Johnson & Johnson vaccine makes up less than 5% of the recorded shots in arms in the U.S. to date,” White House Covid-19 Response Coordinator Jeff Zients said. The U.S. has enough Pfizer and Moderna vaccines to continue the current pace of vaccinations and meet President Biden’s goal, Zients said in a written statement.
South Africa’s medicines regulator, its health minister and researchers are in talks about whether to pause the rollout of J&J vaccines following the U.S. recommendation, Business Day reported, citing Glenda Gray, head of the South African Medical Research council.
Deutsche Bank to Offer Shots to Workers (8:41 a.m. NY)
Deutsche Bank said it’s partnering with Premise Health to begin offering coronavirus vaccines to its employees and eligible dependents in New York on April 20, subject to availability, according to Politico’s Morning Money.
“As far as we know, we’re the first and only Wall Street bank to be offering employees and dependents vaccine appointments from the office,” spokesman Dylan Riddle told MM.
South Sudan Misses Vaccine Target (8:20 a.m. NY)
South Sudan vaccinated less than a fifth of the number targeted in its first week of inoculations because of a lack of transport. The East African nation has only administered shots to 947 health workers, while it planned for 5,000, Health Ministry official George Auzanio Legge said by phone. The ministry now intends to travel to them instead, he said.
Zambia to Start Vaccinating (7:37 a.m. NY)
Zambia will begin its vaccination program on Wednesday using AstraZeneca shots secured through the Covax facility to inoculate about 3.7 million people, Health Minister Jonas Chanda said. The southern African nation, which has to date recorded 90,218 cases and 1,229 deaths, will begin with front-line health workers.
Scotland Lifts Domestic Travel Ban (7:32 a.m. NY)
Scotland’s semi-autonomous government lifted a ban on travel within the country earlier than planned as progress with vaccinations helps reduce new cases, First Minister Nicola Sturgeon said.
From April 16, Scots will be able to travel freely within the country and no longer have to stay within their own local authority area, she told reporters in Edinburgh on Tuesday. The government also confirmed plans to ease restrictions on travel to and from England and Wales from April 26.
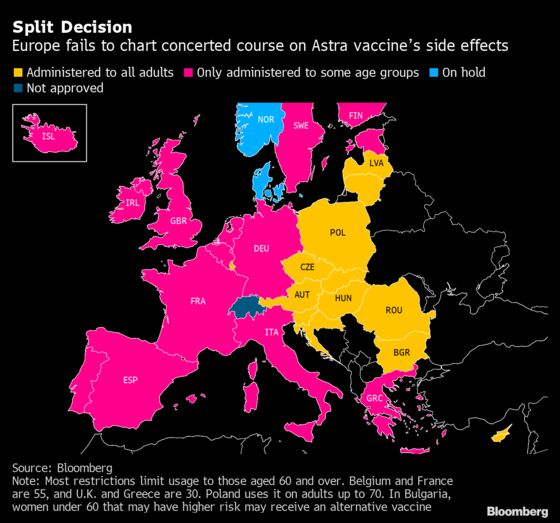
EU Health Chief Seeks States’ Astra Vaccine Data (6 a.m. NY)
Stella Kyriakides, the European Union’s health commissioner, asked member states to provide data on potential side effects from AstraZeneca’s vaccine by Friday at the latest, in order to develop a coordinated approach to restrictions in administering the shot.
She requested the data to allow the agency to better characterize the benefit and risk of the Astra vaccine in different age groups and/or genders, as well as to identify other possible other risk factors, according to a letter seen by Bloomberg.
“Only on this basis, we will be able to ensure a coordinated European approach which does not confuse citizens, and that does not fuel vaccine hesitancy because it is based on robust scientific evaluation,” Kyriakides said in the letter.
The EU’s efforts to get member countries to take a joint position on the Astra vaccine have so far failed to have much effect. Ireland became the latest country to change guidance on the shot, limiting it to those age 60 and over.
©2021 Bloomberg L.P.
With assistance from Bloomberg