Inside Operation Warp Speed’s $18 Billion Sprint for a Vaccine
Inside Operation Warp Speed’s $18 Billion Sprint for a Vaccine
(Bloomberg Businessweek) -- On a late August day in an industrial corner of Baltimore that had been mostly silenced by the pandemic, a red-brick manufacturing plant was buzzing with activity. Deep in the building, in a zone called Area 3, the stainless steel shell of a bioreactor lay on its side, having just arrived from Massachusetts. Employees had begun the task of making the bioreactor operational. Within weeks it would be the center of a production line for coronavirus vaccines.
When the owner of the plant, Emergent BioSolutions Inc., ordered the bioreactor, one supplier said some critical parts wouldn’t arrive until November or December. And so Emergent enlisted the help of Operation Warp Speed, the federal government’s mission to accelerate development of a Covid-19 vaccine. Officials working with OWS, a couple of whom are embedded with Emergent, called the supplier to say the order couldn’t wait, throwing the weight of the government behind the request. “It’s almost like having that Bat-Signal,” says Syed Husain, who heads the company’s contract manufacturing business. “They’ve been a great partner for us.”

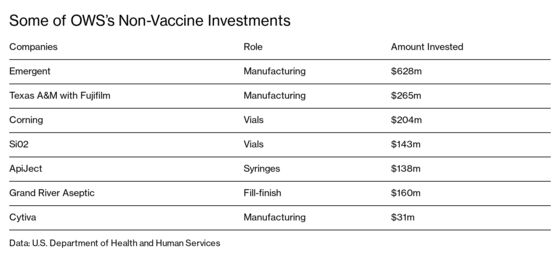
Emergent might be the ultimate Operation Warp Speed company. In June it secured a $628 million no-bid contract to produce vaccines and therapeutics, one of the biggest allocations of OWS funding outside of the pharmaceutical companies actually developing the vaccines. It subsequently signed $261 million in contracts to make doses for AstraZeneca Plc and then a $480 million contract with Janssen Pharmaceuticals Inc., a subsidiary of Johnson & Johnson. Those companies are themselves recipients of billion-dollar-plus OWS awards. Earlier in the year, Emergent produced vaccines for trials by the biotechnology company Novavax Inc., which subsequently received $1.6 billion from OWS. Emergent is in business with three of the six vaccine makers OWS is known to have backed.
Over the past decade, Emergent turned itself into a business the U.S. government couldn’t live without, the sole producer of approved anthrax and single-dose smallpox vaccines stockpiled for emergencies. When the novel coronavirus hit earlier this year and the U.S. government went hunting for surge capacity to make vaccines, it turned to Emergent. “This was actually a kind of global race to secure manufacturing capacity,” says Paul Mango, deputy chief of staff for policy at the Department of Health and Human Services and a key figure at OWS. “We knew we would need an enormous amount of manufacturing capacity to get this done—hundreds of millions of doses in a time that has never been done in history. There wasn’t a lot of idle manufacturing capacity laying around in the U.S.”
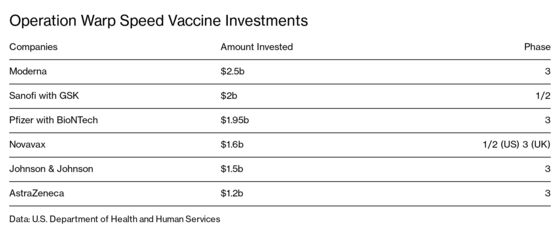
Operation Warp Speed isn’t an agency as such, but rather a mechanism to coordinate among private companies and an array of U.S. government bodies: the Department of Defense, HHS, the Food and Drug Administration, the Centers for Disease Control and Prevention, and beyond. More than 600 people in HHS and 90 people from the DOD are involved. “It’s a coordination activity that helps to cut through the bureaucracy faster,” says Paul Stoffels, chief scientific officer at Johnson & Johnson. OWS has awarded more than $12 billion in vaccine-related contracts and has an overall budget of as much as $18 billion.
As if the logistics and science aren’t challenging enough, President Trump has dialed up the pressure to get a vaccine cleared before the Nov. 3 election. FDA Commissioner Stephen Hahn dashed those hopes in late September when he drew up tough new safety guidelines for approving coronavirus vaccines. Just after he was released from the hospital for his own case of Covid, Trump attacked Hahn by tweet: “Just another political hit job! @SteveFDA.”
The president’s single tangible, constructive contribution to the pandemic response has been to bless the establishment of OWS. Denying the virus was a real threat for months, Trump failed to come up with a national strategy to control its spread and left it to the states to respond. Then he endorsed a company-driven approach. Mango says there are now more than 25 manufacturing sites producing or gearing up to make vaccines, vials, and other things necessary for a mass vaccination program, with OWS tracking progress through streams of spreadsheets.
It appears to be a conspicuous exception to the otherwise disastrous management of the pandemic. “The only part of the pandemic Trump responded to was things he could get companies to manufacture,” says Peter Hotez, dean for the National School of Tropical Medicine at Baylor College of Medicine in Houston. “There was never an understanding that the hard part is giving adequate time to make sure the vaccines work. It was always reframed as a manufacturing problem. It was Trump bringing in his personal relationship with CEOs to get them to make stuff.”

Calling the search for a vaccine Operation Warp Speed hasn’t always engendered confidence. The name comes from Star Trek, and Trekkies know that when a spaceship travels at a high warp factor, things can go wrong. More to the point, Anthony Fauci, director of the National Institute of Allergy and Infectious Diseases (Niaid), has expressed concern that the name suggests recklessness. No one is cutting corners, Fauci insists. Perception issues aside, the idea behind OWS makes sense. A top-notch vaccine specialist from the private sector, former GlaxoSmithKline Plc executive Moncef Slaoui, is paired with military logistics expert Gustave Perna, a four-star general who led the U.S. Army Materiel Command and is now the OWS chief operating officer. They are a study in contrasts: On a recent visit to a Cincinnati hospital taking part in a vaccine trial, Perna was in camouflage fatigues while Slaoui wore a blue button-down shirt.
Guided by Slaoui, a Moroccan-born Belgian-American scientist, OWS selected a portfolio of coronavirus vaccine candidates being developed by some of the world’s top pharmaceutical companies, as well as some untested newcomers. By betting on several horses, OWS in theory increased the chances that one or more would cross the finish line, hopefully at a warplike speed. Slaoui steered investments not just to different companies but also to different ways of making vaccines. Moderna Inc. and Pfizer, which has teamed up with Germany’s BioNTech SE, are testing vaccines that use messenger RNA, a genetic material that instructs the body to make viral proteins that in turn trigger an antibody response—in this case against SARS-CoV-2, the virus that causes Covid. J&J and AstraZeneca are using a viral vector technology that deploys an altered common cold virus to carry the genetic material of the coronavirus and produce an immune response. The Novavax vaccine and one from Glaxo and France’s Sanofi SA are based on the virus’s spike protein and use an immune-boosting adjuvant to spark antibody production. Pfizer might be first out of the gate; the company has said it could be ready to apply for authorization by late November. Unlike the others, the company chose to pay for its own testing and production and will recoup some of its investment only when its vaccine gets authorized and OWS buys the shots.
Being new to government may actually be to Slaoui’s advantage. “Everybody is party to a given kingdom,” he says. “Having somebody from the outside come in that’s effectively blind to all the history and politics, the bureaucracy that may exist, and is uniquely focused on integrating their work … was an essential element of how Warp Speed was able to go very fast.”
OWS released a document in mid-September outlining how it would distribute vaccine doses. It has a military tone; the title is From the Factory to the Frontlines. General Paul Ostrowski, Perna’s deputy, has said the goal is to begin delivery within 24 hours of any approval or emergency use authorization, with distribution centralized and farmed out mainly through McKesson Corp., a Texas drug distributor that had an existing contract with the CDC to deliver vaccines. To track where every dose goes, OWS is building an integrated computer system that brings in the CDC’s existing systems. OWS says it expects to have as many as 75,000 injection sites across the country. States have sent the CDC proposals for how to manage distribution locally, but in mid-October the bipartisan National Governors Association submitted a long list of questions to the Trump administration about everything from funding to storage, making it clear the states don’t see a workable federal plan in place.
Some observers have questioned how a victory for Joe Biden might affect the course of the program. According to Mango, the point is largely moot. The U.S. is on track to produce more than 100 million doses of coronavirus vaccines, manufactured by multiple companies, by the end of the year, he says, and by Inauguration Day, “the vast, vast majority of the heavy lifting will be behind us.” The Biden campaign says it will back scientists involved with Warp Speed. “Joe Biden and Kamala Harris will provide the leadership that has been lacking under Trump to empower scientific professionals throughout our government—including those involved in Warp Speed—to ensure that a safe and effective vaccine is distributed equitably, efficiently, and free to all Americans,” spokesman Andrew Bates said.
Mango says more than 600 million shots should be available by April 2021. Behind those big numbers are a lot of variables and uncertainties. Five of the six OWS-backed vaccine candidates require two doses. It’s possible the FDA will not approve all of them, which means untold millions of doses could end up in the garbage. (And indeed, AstraZeneca and J&J have had to pause their U.S. trials because of safety concerns. Both companies resumed on Oct. 23.) Each vaccine has its own storage requirements. Pfizer’s needs to be kept at -75C (-103F), which makes distributing it across the country a daunting task.

The creation of Operation Warp Speed might have been unnecessary if not for a series of decisions that now look tragically myopic. Public-health experts had been sounding the alarm about the lack of pandemic preparedness for the past two decades, but they were drowned out by concerns over a bioterrorist attack. Those fears were amplified in June 2001 when a group of U.S. officials and policymakers convened for Dark Winter, an exercise which simulated a biological weapons attack involving smallpox. In many ways, the exercise gamed out much of what has unfolded in the U.S. during the Covid pandemic, including the lack of surge capacity to treat victims and the difficulty of controlling the outbreak.
After Dark Winter came Sept. 11 and a series of highly publicized anthrax attacks in the U.S., ushering in a new round of government spending to ward off a bioterrorist attack. Emergent, then called BioPort Corp., was the lone producer of the only FDA-licensed anthrax vaccine. The federal government soon began stockpiling millions of doses.
In 2004, Congress passed the Project BioShield Act, which provided $5.6 billion over the following decade to develop and purchase “medical countermeasures”—vaccines and therapies against biological and chemical attacks—for the Strategic National Stockpile, including Emergent’s anthrax shot. The act wasn’t designed to tackle naturally occurring infectious diseases, despite coming right on the heels of severe acute respiratory syndrome, or SARS, which was caused by a coronavirus. SARS had by that time infected 8,000 people globally and killed 774, with outbreaks still popping up in Asia that year.
Drug companies weren’t eager to invest in the development of drugs and vaccines needed for the Strategic National Stockpile—they’re often risky research bets with an uncertain payoff—so in 2006 Congress created the Biomedical Advanced Research and Development Authority, known as Barda, to fill that void. Through a series of Barda contracts and acquisitions, Emergent consolidated its grip on the biodefense industry. In 2007 it secured a contract to deliver 19 million doses of its anthrax vaccine for the national stockpile. It then acquired and renovated a facility in Baltimore—the one now producing Covid vaccines.
There’s a world in which those vaccines might have come sooner. In 2012, Hotez, the Baylor scientist, got a large Niaid grant to devise a vaccine against SARS. By 2017 the grant ended, and even though the vaccine looked promising, he couldn’t find money to begin testing it in humans. He says it might have provided some protection against the new coronavirus. “We had it in the freezer,” he says. “These are not really moneymaking products.”
President Obama said in an October speech that he left the Trump administration a 69-page document on how to deal with emerging pathogens, including coronaviruses. “We literally left this White House a pandemic playbook,” he said. But the Trump administration has made a habit of pulling the plug on Obama-era initiatives. In 2018, Trump’s national security adviser, John Bolton, dissolved the Directorate for Global Health Security and Biodefense, created by Obama after the Ebola epidemic of 2014 to coordinate the White House response to outbreaks of infectious diseases around the world. Bolton said via Twitter that global health remained a top priority of the National Security Council and that its experts effectively handled the 2018-19 Ebola outbreak.
Responsibility for pandemic planning has long been scattered across the federal government. There’s nothing as visible as a Federal Emergency Management Agency. Perhaps the closest thing is the Office of the Assistant Secretary for Preparedness and Response, headed by Robert Kadlec, a doctor and retired Air Force colonel. A year after being appointed to the position by Trump, Kadlec took control of the $650 million budget for the Strategic National Stockpile, which had been managed by the CDC. Kadlec was at one time a paid consultant for Emergent and had been in business with the company’s founder, the Washington Post reported in May. Asked whether Emergent’s relationship with Kadlec put it in a favorable position, the company said it’s worked with multiple administrations, both Democratic and Republican. Under the Trump administration, Emergent’s revenue has more than doubled, to $1.1 billion in 2019 from $488 million in 2016. When the pandemic hit, the Strategic National Stockpile had millions of doses of anthrax vaccine and enough smallpox vaccine to inoculate every U.S. citizen.
It’s not as though Kadlec didn’t recognize the risk of a naturally occurring pandemic. In 2019 his office oversaw a simulation of an influenza pandemic emanating from China. The exercise, called Crimson Contagion, laid bare how unprepared the U.S. was for something like the novel coronavirus. In testimony before Congress in December 2019, Kadlec said the U.S. lacked sufficient manufacturing capacity for almost all necessary pandemic supplies: vaccines, therapeutics, needles, syringes, and masks. If the simulation was designed to spark significant changes to the government’s planning, they didn’t come in time.

On Jan. 13, Sean Kirk, Emergent’s head of manufacturing and technical operations, grew worried as he watched the news about a respiratory illness spreading in Wuhan, China. He walked down the hall to his colleagues running the company’s global supply chain and told them to start locking up critical raw materials. “I was fearful that this thing could blow up,” he recalls. Later that month, Emergent submitted an in-depth white paper to Barda reminding the agency of everything the company could do.
Rick Bright, who was serving as director of Barda as the virus hit the U.S., filed a whistleblower complaint earlier this year that painted a picture of a federal government that was slow to act on vaccines. He says he was ousted in April after fighting pressure to allow use of hydroxychloroquine, an anti-malaria drug that Trump had been pushing despite the lack of rigorous trials to prove it was safe and effective. In January, Bright says, he tried and failed to persuade Kadlec and HHS Secretary Alex Azar to allocate money for Covid drug and diagnostics development. In a written statement, the HHS said that from his position, Bright couldn’t know everything being done to combat the pandemic. Bright didn’t respond to requests for comment for this story.
Barda pressed ahead on Covid anyway. In January the agency contacted Johnson & Johnson to discuss using the same approach for the novel coronavirus that it had used for its Ebola vaccine, recalls Stoffels, the chief scientific officer. “We know the technology you have is developed and you can scale it,” he recalls Barda experts saying. Over three meetings in Washington in February, Stoffels says, the company and Barda came up with a development plan. As it hammered out the initial contract, J&J began talking to Emergent about locking up manufacturing capacity. On March 30, HHS announced $456 million in funds to support clinical trials and manufacturing of J&J’s eventual candidate, followed by an additional $483 million for Moderna. In April, Emergent signed an initial $135 million agreement to manufacture J&J’s vaccine, with some work starting in 2020 and large-scale production beginning in 2021.
By mid-April, daily deaths from Covid were surging, and the pressure to act was growing. HHS officials were fleshing out how to set up OWS. The decision to create a new body struck some as an unnecessary gimmick, given there were existing interagency structures in place that the government used to respond to Ebola.
Although OWS wasn’t formally announced until mid-May, HHS and the DOD started laying the groundwork by interviewing as many as 10 people for the role of chief adviser. Azar, Defense Secretary Mark Esper, and White House adviser and Trump son-in-law Jared Kushner interviewed Slaoui and decided he was the right person for the job. He joined OWS as an outside contractor instead of as a government employee, which meant he wasn’t subject to federal ethics rules regarding disclosure and divestment of shareholdings that might conflict with his role. Slaoui agreed to work for OWS for a nominal $1 salary, with his expenses covered under an HHS contract awarded to Advance Decision Vectors LLC, based in Alexandria, Va., which is handling compensation for several OWS consultants.
Slaoui has come under fire from congressional Democrats, who say he has too many conflicts of interest with OWS companies. He vociferously denies that. (He says he’s a registered Democrat who joined OWS because he believed finding a vaccine was more important than party politics.) After heading vaccines at GlaxoSmithKline, he’d taken board seats at Moderna and, briefly, the Swiss drug manufacturer Lonza Group Ltd., Moderna’s vaccine maker of choice. When he agreed to join OWS, he resigned from Lonza and Moderna and promised to sell all his Moderna shares. He insisted on keeping his multimillion-dollar stake in Glaxo, which he’s called his “retirement,” but pledged to donate any gains to the National Institutes of Health from the day he started at OWS. He says he wasn’t involved in negotiating the deals with Moderna or the Sanofi-Glaxo partnership. “I stayed completely out of it,” Slaoui says.
On July 2, Francis Collins, the NIH director, and Gary Disbrow, acting Barda director, testified on OWS before Congress but refused to provide details on how the program would pick vaccines to fund. At that point, OWS had announced $1.2 billion for AstraZeneca’s vaccine developed with the University of Oxford. Democrats asked how many vaccines it would back. “More than one,” Disbrow said with a chuckle. “I’m sorry—it really is procurement sensitive.”
Over the next six weeks, OWS handed contracts worth more than $8 billion to back five additional vaccines—those from Johnson & Johnson, Moderna, Novavax, Pfizer, and Sanofi jointly with Glaxo. The government reluctantly released some of the contracts—heavily redacted—over the summer in response to Freedom of Information Act requests. Slaoui says the fuss is unnecessary: “They are plain-vanilla contracts. There’s nothing special about them apart from the amount of money, which relates to the cost of developing the vaccines.” In mid-October, the advocacy group Public Citizen filed a lawsuit demanding the release of coronavirus vaccine contracts. In late October, HHS released a Moderna contract, heavily redacted.
OWS deals have led to a huge payday for many pharma executives. Moderna executives netted a profit of $115.5 million from selling shares in their company from May 15 to Aug. 31, according to Accountable Pharma, a nonpartisan watchdog group. A Moderna spokesman said the share sales were done through previously scheduled programs and in accordance with insider-trading regulations. Emergent executives netted $5 million selling shares during the same period, and in September the company’s executive chairman, Fuad El-Hibri, sold an additional 80,000 shares for a net profit of $8.1 million. Emergent said in a statement that executives regularly sell stock, following all laws governing financial transactions. Emergent’s shares were up 78% this year through Oct. 23.
“It’s a perfect business,” says Eli Zupnick, a spokesman for Accountable Pharma, of the OWS companies. “Their downside is covered by taxpayers, and their upside is already in their pockets.”

After announcing the $628 million OWS award for Emergent in June, Perna and Slaoui visited the company’s Baltimore manufacturing facility to deliver a message that the entire U.S. military was ready to help work out supply chain glitches. “He had said that everything is on the table,” Emergent’s Husain recalls of Perna’s message. “Every day counts, every hour counts.”
Some pharma executives describe the military’s role in speeding up vaccine manufacturing as a genuine bright spot in the federal pandemic response. Much of the work, however, remains opaque. Ostrowski, Perna’s No. 2, says OWS intervened on behalf of suppliers working with Moderna to expedite orders of key equipment and supplies, but he didn’t offer any specifics. “My hat goes off to these great Americans that want to be part of the solution to this national pandemic,” he said in emailed responses to Bloomberg Businessweek’s questions. Moderna is the largest recipient of taxpayer funds—a total of $2.5 billion—to advance and supply its vaccine, including a $300 million milestone payment if it’s able to deliver 100 million doses by Jan. 31, 2021, under an FDA emergency use approval or full license. Moderna said in a statement that it interacts with several federal agencies but declined to say what support it’s getting. The milestone payment didn’t require the company to accelerate timelines for its trials, a spokesperson says.
Amid widespread fears there would be a shortage of key materials for a mass vaccination program, OWS signed contracts to finance large-scale manufacturing of hundreds of millions of vials. Corning Inc. secured $204 million in funds from OWS in June to expand its capacity to produce specialized glass vials. The company has weekly calls with OWS and has used a system under the Defense Production Act to gain priority with suppliers. “It’s been a helpful tool to make sure we’re not getting stuck in line behind anything that’s noncritical,” says Brendan Mosher, Corning’s head of pharmaceutical technologies. “We’re doing things at least three times faster than we would have.”
Not every company is treating its relationship with OWS like a military secret. SiO2 Materials Science of Auburn, Ala., won a $143 million award in June to produce 120 million plastic vials with a thin glass coating for Moderna’s vaccine and several others that it declined to disclose. Lawrence Ganti, SiO2’s president of customer operations, says the company has used the Defense Production Act to solve myriad problems to keep production humming. Over the summer, one of its suppliers outside Chicago shut down because of a local power outage. Ganti says he got the power company’s head of legal affairs on the phone, gave her the supplier’s Defense Production Act letter, and got the power restored within 45 minutes. “They understood the gravity of what we were working on,” he says.
Mango says he’s confident the U.S. will be ready to begin vaccinating millions of people by the end of the year. “The vaccines will be ready on the shelf,” he says, declining to say which vaccine he’s referring to. “We’ll have all the needles, syringes, stoppers, and vials—all of that stuff will be ready to ship literally the day the FDA gives us a green light.”
If Emergent is anything to go by, that timeline looks like a stretch. Its first doses of the AstraZeneca vaccine won’t be available until late January at the earliest. Production finally started in mid-September, but it will take 90 days to produce bulk doses, which will then be shipped to a separate company, Catalent Inc., based in Somerset, N.J., for filling into vials, a process that takes another 45 days including quality-control checks. Fauci confirmed in late October that vaccines won’t be available in the U.S. until January, if then.
With so many vaccines in the race, OWS can turn to others if the candidates made at Emergent don’t cross the finish line, but fewer approvals would diminish the chances of delivering millions of doses quickly. The world needs more than one shot to get back to normal. “Success for Operation Warp Speed is not six vaccines all working. It’s enough vaccines in enough quantities by yearend to vaccinate the most vulnerable Americans,” Mango says. “The odds of six out of six hitting in the time frame we want, I mean, you’d sooner get struck by lightning.”
The biggest threat to OWS’s success may end up being not the science, not the logistical challenges, but people refusing to take an approved vaccine. A Pew Research Center study in September showed that just 51% of U.S. adults say they would definitely or probably take it—down from 72% in May.
“I think this is a result mostly of the politicization of this process and the fact that it’s happening around an election year and the kind of politics that are going on in the country,” Slaoui says. His hope is that 60% to 70% of Americans will be immunized, at which point, though Covid won’t disappear, the pandemic will subside. “The circulation of the virus will be hugely impaired,” he says. “Infections will go down, susceptible people will survive, and life will go back to normal. I look forward to that.” —With David Kocieniewski
Read next: Better, Faster Testing Is the Path to an American Comeback
©2020 Bloomberg L.P.