UniQure Sinks While Gene Therapy Study Pause Casts Pall on Peers
UniQure Sinks While Gene Therapy Study Pause Casts Pall on Peers
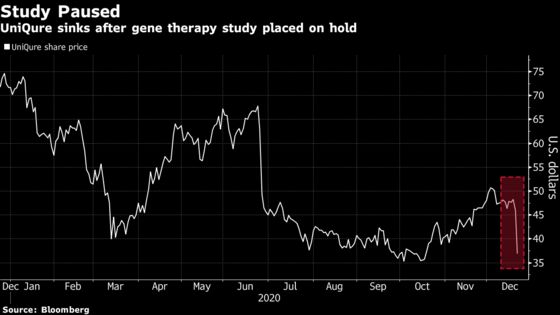
(Bloomberg) -- UniQure NV plunged in Monday trading after its hemophilia B gene therapy program was placed on clinical hold by U.S. regulators over safety concerns.
Other companies working on similar medicines followed the biotech lower. Shares of BioMarin Pharmaceutical Inc., Rocket Pharmaceuticals Inc. and Solid Biosciences Inc. all fell in the low- to mid-single-digits, while UniQure fell as much as 21%, the biggest drop since late June.
The U.S. Food and Drug Administration put the program on pause after one patient of advanced age who was treated with the experimental therapy etranacogene dezaparvovec in October of last year received a preliminary diagnosis of hepatocellular carcinoma.
The patient had multiple risk factors associated with liver cancer, including a 25-year history of hepatitis C, hepatitis B and evidence of non-alcoholic fatty liver disease, the company said in a statement.
“We do not anticipate any impact to our regulatory submission timeline for the hemophilia B program as a result of this clinical hold,” the company’s chief executive Matt Kapusta said.
©2020 Bloomberg L.P.