Moderna, BioNTech Rally Stalls While CDC Meeting Weighs Boosters
Wall Street waits for CDC to weigh in on whether Americans should get a third shot.

(Bloomberg) -- The huge rally for Covid-19 vaccine makers has stalled as Wall Street waits for the U.S Centers for Disease Control and Prevention to weigh in on whether Americans should get a third shot.
On Monday, a meeting of outside advisers to the agency could finally give investors a sense of clarity. Advisers were convening to discuss the framework for a third jab in the late afternoon following a unanimous vote recommending Pfizer Inc. and BioNTech SE’s shot for people of 16 and older.
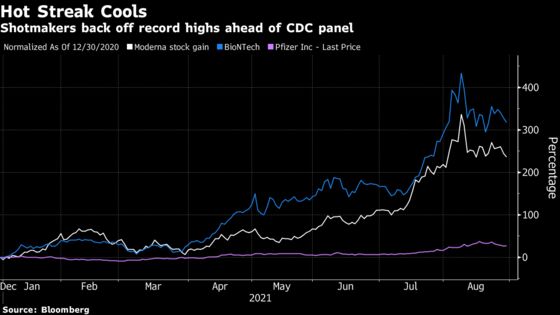
Moderna Inc., which became the best performer in the S&P 500 Index after its stock price more than tripled this year, has slid 23% from its record high in early August. BioNTech SE, which has surged more than four-fold in 2021, has fallen about as much as Moderna over the past three weeks, while its U.S. partner, Pfizer Inc., is off about 7% from its peak.
The retreat comes as the pace of the American inoculation campaign slows, leaving investors divided over how much growth to expect from the manufacturers. That rift has only deepened after President Joe Biden’s administration promised that booster shots will be made available to most Americans, which would be a major source of demand if regulators endorse that step.
The administration’s aim to push through new guidelines in late September, however, has been met with skepticism by some medical experts. The CDC as well as the U.S. Food and Drug Administration have to approve the plan before it can be implemented and that backing isn’t guaranteed.
Shares of Moderna were also under pressure Monday from a U.S. regulatory warning on heart risks and a Japanese probe into the company’s shot following the deaths of two men and the suspension of three lots of the vaccine.
That uncertainty is likely to continue fueling volatility in vaccine-makers. Options imply that Moderna and BioNTech could move roughly 6% in either direction this week while Pfizer’s stock may move about 2.4%.
Morgan Stanley’s Matthew Harrison is among Wall Street analysts who expect Biden’s plan to be backed by regulators. But but he remains equal weight on Moderna and Pfizer because he doesn’t expect any major new vaccine orders in the near future, with an estimated 630 million of unused shots from the companies already purchased through 2022.
“We do not expect any supply constraints for booster doses and do not expect the U.S. government to purchase additional mRNA vaccine doses for 2022 based on current demand or booster dose need,” Harrison wrote in a research note, referring to the technology used in both Moderna and Pfizer’s shots.
Analysts with Goldman Sachs Group Inc. are more bullish. The bank has a buy rating on Moderna, estimating that its shot could drive $36.2 billion of sales next year, some $15 billion more than Bloomberg’s consensus estimates. “We see upside to these numbers given supply and pricing dynamics,” said analyst Asad Haider.
He sees new records for Moderna and Pfizer if U.S. and other global regulators endorse the receipt of a third dose. He estimates that Moderna shares -- which closed Friday at $382.22 -- could top $600 if boosters become the norm, while Pfizer could trade as high as $61, up from $46.58 at the end of last week. BioNTech already trades at best-case levels, he wrote.
On Monday, Moderna slid 3%, BioNTech fell 3.8% and Pfizer ended the day little changed.
©2021 Bloomberg L.P.