Miracle Cancer Drugs Are Making Big Pharma Billions. Others Are Getting Left Behind
Miracle Cancer Drugs Are Making Big Pharma Billions. Others Are Getting Left Behind
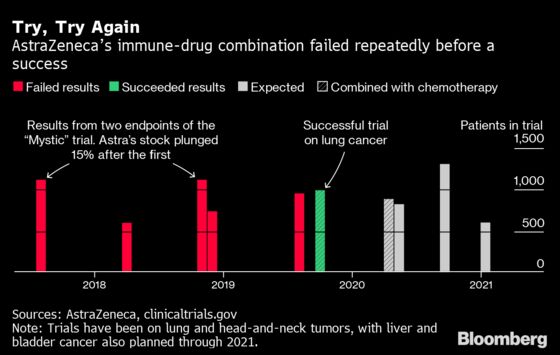
(Bloomberg) -- It looked like a massive bust. In one painstaking test after another, AstraZeneca Plc’s combination cancer therapy failed. One of the final-stage trials tanked the U.K. drugmaker’s shares and appeared to doom the vaunted treatment’s prospects.
But Astra pressed on, spending millions of dollars to try treating thousands of patients. The series of disappointments finally ended in October, when adding the drug combination to standard chemotherapy for lung tumors yielded a ray of hope.
Dazzled with a treatment revolution that puts cures within reach for some of the most stubborn cancers, pharma companies are willing to stretch their resources like never before—a startling drift in an industry that prides itself on knowing when to quit on dead-end drugs.
Merck & Co. and Sanofi on Monday clinched deals with hefty premiums to stake their claims in the increasingly crowded cancer space. The French drugmaker went one step further, ending research in its legacy areas of diabetes and heart disease.
“Our goal is to eliminate cancer as a cause of death,” Dave Fredrickson, head of drug development for Astra in the field, said in October. The company’s dogged support for a combination treatment has faced questions since 2017, when a crucial test called Mystic in lung cancer failed.
The rewards are eye-popping: Merck’s Keytruda, the leader in a class of medicines that harness the immune system, could attain annual sales of more than $27 billion within six years, according to estimates, more than any drug in history. Lifted by the rising tide of new treatments, the global oncology market is expected to reach $230 billion by 2024.
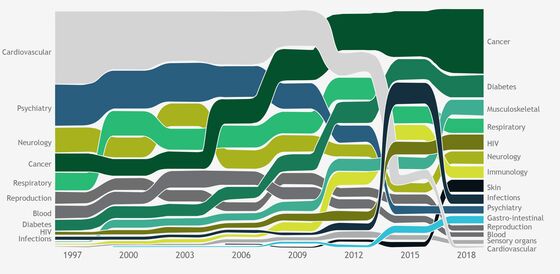
The cancer scramble comes at the expense of conditions like multiple sclerosis, psoriasis, asthma, infectious diseases—and even opioid addiction—that have been less lucrative for pharma companies. Oncology therapies’ share of U.S. drug revenue almost quadrupled to 28% over the past two decades, even as cardiovascular drugs dropped from dominance to 1%, according to Boston Consulting Group.
“No company has an infinite amount of money, so they’re having to make choices,” said Janice Reichert, executive director of the Antibody Society, a Massachusetts-based trade group.
Not that cancer is an unworthy target. It ranks as one of the world’s lethal ailments after medical advances eradicated many others. The disease is to blame for 9.6 million deaths last year, making it the second-leading killer globally, according to the World Health Organization. It remains “an impossible challenge for patients and families,” said Jay Bradner, head of Swiss drugmaker Novartis AG’s research arm.
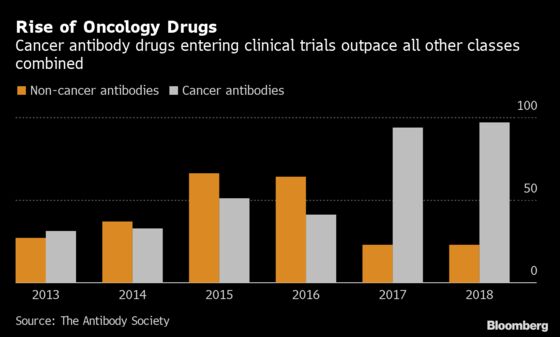
A better understanding of the disease’s biology—starting in the 1970s with the war on cancer, through the Human Genome Project and immune-oncologystudies that won Nobel recognition—is feeding the revolution. Some 3,900 immune-oncology treatments are now in different stages of testing as companies pan for new products.
“There’s some emotion around cancer that drives a lot of spending,” said Lars Fruergaard Jorgensen, the chief executive of the Danish drugmaker Novo Nordisk A/S, which has chosen not to invest in the field. “In my book, there’s way too much investment going into oncology compared to what comes out of it.”
Spurring the race are reimbursement prospects. Drugmakers are increasingly wary of products that U.S. payers may spurn, and unlike diabetes or heart medicines, cancer treatments gain relatively swift endorsement, said Steven Nissen, chairman of cardiovascular medicine at the Cleveland Clinic in Ohio.
“We can’t save lives unless there’s innovation,” he said in an interview, “and you can’t have innovation unless someone’s willing to pay for it.”
Drugmakers notice which products deliver the best returns.
After spending $8 billion to buy Loxo Oncology in January, diabetes drugmaker Eli Lilly & Co. said last week it would expand its global presence in cancer, hiring Memorial Sloan Kettering Cancer Center’s David Hyman as chief medical officer of a reorganized oncology division.
Pfizer Inc., maker of Ibrance for breast tumors, is hiving off its generic-drug division while adding cancer emphasis with the June purchase of oncology biotech Array BioPharma Inc. for $11.4 billion.
GlaxoSmithKline Plc has been the U.K.’s biggest health company by revenue for decades, specializing in asthma and HIV therapies. Over the past two years, its market value dropped behind Astra’s as a lung cancer medicine called Tagrisso fueled the smaller drugmaker’s sales.
In catch-up mode, Glaxo last year paid about $5.1 billion for Tesaro, the maker of a promising cancer medicine called Zejula. An oncology partnership with Germany’s Merck KGaA, worth as much as $4.2 billion, followed earlier this year. Chief Executive Emma Walmsley has doubled the number of experimental cancer drugs in human testing to 16, including three that could reach the market next year. At the same time, the company has halted dozens of other programs.
The cancer focus has rekindled Astra. Five years ago, CEO Pascal Soriot didn’t see oncology as a major contributor to growth. Now the company has three cancer drugs that will be blockbusters this year and at least one more in waiting, making it a major player in the field. Astra has also recently developed drugs for diabetes, heart and kidney disease, as well as other, less common conditions.
Astra is in cancer “because we have the right science,” Soriot said in an interview. “You can’t say you’re going to be in the field because there’s money to make. If you don’t have the products, you can’t get there.”

In come cases, it’s been an uphill battle. Astra’s stock plunged 15% the day the day its combination of the experimental drug tremelimumab with Imfinzi, an approved treatment, failed the key Mystic trial.
The medicine went on to deliver four more failing results in final-stage tests over the past two years. Finally, in October, a trial showed that adding it to chemotherapy slowed lung-cancer growth. Yet it’s still unclear whether the medicine will ever be submitted for regulatory approval. Meanwhile, four more tests will report results over the next two years.
Only about 1 in 20 cancer patients participates in trials of experimental therapies. Doctors and drugmakers should be prudent about continuing to test products in desperate, hard-to-find patients when they haven’t shown significant benefits in multiple earlier studies, said Jonathan Kimmelman, a biomedical ethicist at McGill University in Montreal.
“You have a ridiculous number of drugs and targets and combinations you can test,” said Kimmelman, director of the Studies of Translation, Ethics and Medicine research group. “You want to pull the weeds and take the drugs and combinations that have the best prospects.”
Proving that a drug works can take time. Failure against one type of cancer doesn’t mean a treatment won’t work in another, said Janet Woodcock, director of drug evaluation for the U.S. Food and Drug Administration. And patients with few or no options may be eager to try an experimental drug regardless of its history.
At least one other Astra cancer medicine was rescued from the jaws of defeat. Not long after Soriot took the reins, he jump-started a halted project that has since become Lynparza, a class leader that treats breast and ovarian tumors. Another candidate in co-development with U.S. drugmaker Merck, selumetinib, failed in a series of human tests before showing promise in a rare childhood cancer, and has been submitted for approval.
“If we have some trials that fail, so be it,” said Jose Baselga, the former chief medical officer of Memorial Sloan Kettering hired to head Astra’s cancer research. “It’s the right thing to do.”
--With assistance from Jeremy Diamond.
To contact the editor responsible for this story: Eric Pfanner at epfanner1@bloomberg.net, Marthe Fourcade
©2019 Bloomberg L.P.