Inovio Tumbles After Analyst Warning Ahead of Vaccine Data
Inovio’s 847% Surge Spurs Analyst Warning Ahead of Vaccine Data
(Bloomberg) -- Inovio Pharmaceuticals Inc. dropped as much as 21% after a Stifel analyst cut his rating following a nine-fold increase, driven in part by day traders’ enthusiasm for small-cap Covid-19 plays.
“Risk/reward seems less palatable here,” Stephen Willey wrote, downgrading the biotech company to hold from buy. Hope and sentiment have replaced valuation, he said, though he acknowledged “the significant socioeconomic importance of a viable Covid-19 vaccine candidate.”
Investors are expecting a first look at results for Inovio’s vaccine candidate, INO-4800, in patients by the end of the month. As the U.S. stock market awaits new treatments and a potential shot to ward off the pandemic, the popularity and shares of vaccine and drug developers have swelled. Inovio, alongside another vaccine name, Vaxart Inc., is one of the hottest day-traded stocks in the past 24 hours according to RobinTrack, a service that follows Robinhood trading.
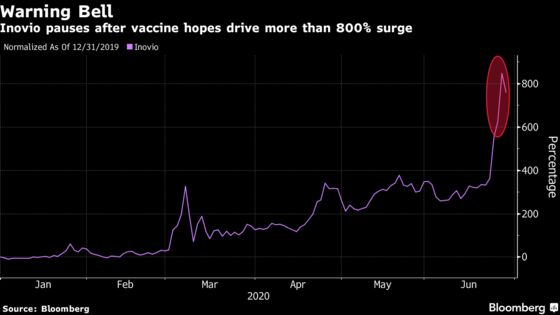
Most of the move ahead of the results may have happened already as options data imply a 35% move by July 2. Inovio was up 847% year-to-date at the close yesterday.
Safety shouldn’t be an issue in Willey’s estimation, and he expects initial immunogenicity results to be “directionally encouraging.” He does see an issue in how it measures up compared to an inoculation from one of the leading developers in the space, Moderna Inc.
The neutralizing antibody results from Moderna’s vaccine “represent a challenging competitive hurdle,” he wrote in a note to clients. Moderna’s first look at a handful of patients showed they had antibody levels similar to those who had successfully fought off infection from the coronavirus. Data from Inovio’s other vaccines in development may mean longer follow-up is needed to show a significant effect.
Price Target
At the same time Willey counseled caution on Inovio’s stratospheric rise, he raised his price target to $24 from $19. Like other analysts, he has had to increase targets to keep up with the stock. Cantor Fitzgerald analyst Charles Duncan upped his price target to $45 from $17 on Friday morning, while keeping his overweight rating on the stock.
Another concern will be Inovio’s ability to scale-up production of a potential vaccine. It is currently in a dispute with the primary manufacturer of the shot, South Korea-based GeneOne Life Science Inc. and its VGXI unit. The Plymouth Meeting, Penn.-based company’s request for a preliminary injunction against VGXI was denied by a judge on Thursday, Philadelphia Business Journal reported. Willey said he expects a near-term resolution of the dispute.
Investors are also waiting for “a large government-written check,” Willey said. There is more than $10 billion in funds rumored to be earmarked for dissemination by a number of different agencies/entities, Vaxart may be one of the latest winners after its vaccine was selected for testing in primates. While a recent Department of Defense contract for Inovio may signal more funding is coming, it remains one more source of uncertainty, according to the analyst.
©2020 Bloomberg L.P.