European Regulators Approve Sales of First Artificial Heart
Shares in Carmat SA posted their biggest gain after the company got approval to sell the first-ever artificial heart in Europe.

(Bloomberg) -- Shares in Carmat SA posted their biggest gain in more than seven years after the company got approval to sell the first-ever total artificial heart in Europe, the culmination of a 27-year effort that began with a pitch from a French cardiac surgeon to an aerospace company.
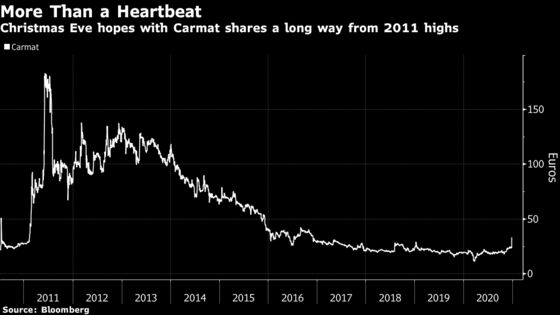
The stock climbed 34% to 32.05 euros at 11:29 a.m. in Paris after rising as much as 50%, its steepest intraday gain since May 2013. Carmat shares have advanced 66% this year, giving the company a market value of about 407 million euros ($496 million).
Carmat this week obtained a CE mark for the device, meaning the company can now sell it in the European Union as a bridge to a heart transplant for patients suffering from irreversible end-stage heart failure.
Surgeon and heart-valve inventor Alain Carpentier proposed development of an artificial heart to French industrialist Jean-Luc Lagardere in 1993. Lagardere put a few labs and engineers from his Matra missile company at Carpentier’s disposal and the project expanded over the years, including through the mergers that led to Matra becoming part of what is now Airbus SE. The planemaker is Carmat’s biggest shareholder, with a 13% stake, while Carpentier owns about 5.3%.
Read more: First Artificial Heart Joins Aerospace With Calf Tissue
It took 10 years for Carmat to obtain the CE mark, and “it’s a record, given the complexity of such a device,” Carmat Chief Executive Officer Stephane Piat said on Thursday in an interview with BFM Business television. “We’ll have to work with doctors and medical centers now to offer our therapy and we’ll have to look for patients. The production phase will be a delicate one.”
Carmat plans to ramp up manufacturing of the device in January with a second shift, said the CEO, who took the helm four years ago.
Carmat’s artificial heart has blockbuster potential and could reap 700 million euros in annual sales in Europe by 2030, Portzamparc analyst Mohamed Kaabouni wrote in a note on Thursday.
“Everyone was waiting for it, and it’s here now,” Kaabouni wrote about the Carmat heart. “The CE mark is crucial for patients and the French company probably got approval for a blockbuster, given how large the medical need is and how rare and ineffective other solutions are.”
Carmat will start selling the device in the second quarter, the company said in Wednesday.
©2020 Bloomberg L.P.