Drug Crafted From Deadly Bacteria Is Changing Kidney Transplants
Drug Crafted From Deadly Bacteria Is Changing Kidney Transplants
(Bloomberg) -- Delilah Romero was only 23, and it felt like her life was over.
When her kidney transplant failed in 2011, her immune system developed an exceptionally high level of antibodies to fight foreign tissue, leaving her sensitized to donated organs. Her chances of finding a replacement her body would accept plummeted to less than 1 in 1,000.
“Would I ever get a transplant? I thought I was going to be on the waiting list forever,” she said from her home in Pasadena, California.
Doctors believe they’ve found an answer for patients like Romero in a protein that is produced by lethal bacteria. The protein, which temporarily wipes out antibodies, was crafted into an experimental drug called imlifidase to give donated organs a fighting chance against the immune system’s defenses.
Developer Hansa Medical AB says imlifidase could make transplants possible for about 35,000 U.S. patients who currently have poor odds, and increase matches for others.
“This really has changed the game,” said Stanley Jordan, a transplant surgeon at Cedars-Sinai Medical Center in Los Angeles who’s run studies of the drug.
With soaring rates of obesity and diabetes, the need for new kidneys has risen faster than organ availability over the past two decades. Some patients become sensitized after losing a first transplant, like Romero, or being exposed to foreign tissues through blood transfusion or pregnancy. Their bodies increase defenses, making it harder to find a viable match.
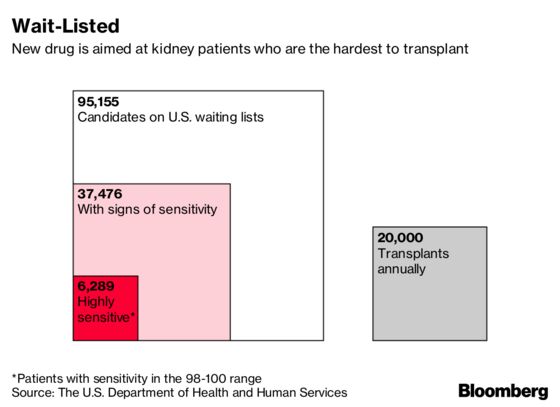
Such sensitized patients account for a third of 95,000 people currently on kidney waiting lists in the U.S. A subgroup of 6,300 are highly sensitized, incompatible with more than 98 percent of organs. For comparison, only about 20,000 U.S. patients get a kidney transplant annually.
‘Matchability of Zero’
In the U.K., only about 3,500 kidneys become available each year. There’s almost no chance of finding a match for patients like Matthew Herbert, a 38-year-old chef in Hampshire, England. His father gave him a kidney, but it was killed off in weeks by his immune system.
“I’ve been told that I have a matchability of zero,” said Herbert, who has been on dialysis for 20 years.
Imlifidase may give patients like him a lifeline. The drug, which is delivered intravenously, saws through the center of Y-shaped antibodies, wiping them out for a couple of weeks. By the time the body starts making new antibodies, the patient is taking immune suppressants and able to hold off organ rejection. Among 35 highly sensitized patients treated in a study, 32 still had working kidneys six months after surgery, according to data released Sept. 27.
Maria Isabel Padilla, 48, is one of them. She was diagnosed with kidney disease in her home country of Mexico age 8 and unable to receive an organ -- even after moving to the U.S. in 1992. After about a quarter-century on dialysis, she got a transplant two years ago following treatment with imlifidase in a clinical trial. Having a healthy kidney is a miracle, she said.
Blockbuster Potential
Hansa Chief Executive Officer Soren Tulstrup says imlifidase has blockbuster potential, meaning annual sales topping $1 billion. The drugmaker plans to file for approval in the U.S. this year or early next. Both U.S. and European Union regulators have granted Hansa eligibility for tax breaks and other incentives for costs of developing treatments for rare conditions.
“The commercial opportunity is significant,” Tulstrup said.

The drug’s journey started a couple of decades ago when Lars Bjorck, a scientist at Sweden’s Lund University, noticed that a human immune protein was missing when a certain strain of bacteria grew in the same samples. Something had destroyed it, quickly and efficiently, and Bjorck determined that the source was a germ called Streptococcus pyogenes. The bacterium, if untreated, can cause deadly flesh-eating infections.
Bjorck isolated the agent in 2001 and found that just one molecule can cripple 2,000 to 2,500 antibodies. For bacteria, that’s like having a light saber to repel an immune attack.
Earlier in his career, Bjorck had experimented with ways to control immune responses to transplants. He immediately visualized a use for his discovery: In the hands of doctors, the protein could temporarily protect organs from being attacked by antibodies.
“I remembered there were quite a number of patients who couldn’t get a kidney because of immune problems,” he said.
A 2014 change in U.S. organ allocation policy has given sensitized patients higher priority. But the current protocol often involves removing anti-transplant proteins from their blood in procedures that can cost as much as $60,000. It can take weeks, so transplants must be scheduled far in advance, which usually isn’t possible with deceased donors.

Few Alternatives
Hansa, based in Malmo, Sweden, is breaking into a field where there are few alternatives, said Roberto Cacciola, a surgeon at the U.K. National Health Service’s Blood and Transplant division.
Laboratories are developing other potential anti-rejection drugs as well as implanted devices and artificial organs designed to do the kidney’s blood-cleaning work, Cacciola said. Researchers are also investigating tissues that are genetically altered to make them compatible to human recipients. But nothing is near becoming an actual treatment.
“We are far from having anything like this available at the moment,” Cacciola said.
Hansa had its own share of hardship. Co-founder Bo Hakansson was killed in 2014 in a motorcycle accident. And in November last year, then-CEO Goran Arvidson died suddenly three months after the company got its breakthrough, the publication of a mid-stage trial of imlifidase in the prestigious New England Journal of Medicine. Arvidson was a kidney transplant patient himself.
‘Very Passionate’
“He was very passionate,” said Emanuel Bjorne, who had been CEO before Arvidson and stayed on as head of business development. “He could relate to all aspects of the opportunity in transplantation, and understood that there’s potential beyond that.”
In March, the company brought in Tulstrup, a veteran of Shire Plc and Swiss drugmaker Vifor Pharma AG who had worked with rare-disease treatments. The new CEO has begun hiring executives with regulatory and marketing expertise to prepare for imlifidase’s run for approval.
The treatment’s arrival on the market would be welcome news for people who depend on hours of weekly dialysis, with the fatigue and depression that often come with it. Romero, the California patient, entered a trial run by surgeon Jordan at Cedars-Sinai and received a new kidney in 2016. Without the need for dialysis, she can travel again and works full-time in health-information management.
“I’m very cautious, but I live a normal life,” said Romero, now 30. “It’s pretty amazing how a kidney can change somebody’s whole perspective.”
To contact the editor responsible for this story: Cecile Daurat at cdaurat@bloomberg.net
©2018 Bloomberg L.P.