Culture of ‘Bending Rules’ in India Challenges U.S. Drug Agency
The FDA confronts creative improvisation in the world’s largest generic-drug exporter.

(Bloomberg) -- Perched on the edge of a sheer rocky outcropping, Mylan NV’s flagship pharmaceutical factory looms over a patchwork of farmers’ fields like one of the medieval hill forts that dot the landscape in this part of India.
Mylan is the world’s second-largest manufacturer of generic drugs, and though it’s run from Canonsburg, Pennsylvania, its operations three hours inland from Mumbai exemplify the central role India has come to play in the global generic-drug industry. About half of Mylan’s workforce is based in India. The company’s president rose through the ranks of its Indian unit. And that hulking, green, glass-and-concrete complex mounted on the hill is one of Mylan’s largest factories.
One of the drugs made in that plant is destined for the U.S., where it’s the second-best-selling generic version of Lipitor, taken by millions of Americans to control cholesterol and lower their risk of heart attack. But a review of thousands of reports submitted to the U.S. Food and Drug Administration shows Mylan’s version of Lipitor is more likely to be associated with negative side effects than its rivals: 60 percent more than the top-selling generic version made by a Canadian company and quadruple that of the third-best-selling generic made by an India-based company, according to the reports from doctors, patients and pharmacists.
The so-called adverse events listed in the documents, obtained through a public-information request, include diarrhea and muscle spasms. For some users, the drug didn’t work at all. And one report about an 82-year-old patient listed the adverse event as death.
The FDA didn’t comment specifically on the reports but cautioned that they don’t all indicate a causal relationship to the drug. “Looking only at the number of AE reports for a particular drug does not give a complete picture of its risk profile, compared to other drugs,” said Sarah Peddicord, a spokeswoman for the FDA.
A yearlong investigation by Bloomberg News into the generic-drug industry shows FDA inspections at factories from West Virginia to China have found reason to doubt the data meant to prove that drugs made there are safe and effective. The Mylan plant in India is just one of the places inspectors say they’ve uncovered such alleged problems. But the persistence of these doubts around the world hasn’t slowed the FDA’s push to bring a record number of new generics to pharmacy shelves in America, as it tries to deliver on President Donald Trump’s promise of lower drug prices.

India is the world’s largest exporter of generic drugs, making almost 40 percent of all new generics the FDA approved in 2018 through October. The FDA has found cause for alarm over the years at Indian factories across companies and around the country, from open toilet drains found at a sterile facility owned by Mumbai-based Wockhardt Ltd. to malfunctioning equipment at one of Dr. Reddy’s Laboratories’ plants in south India. Since then, a Dr. Reddy’s spokesperson said, the company has strengthened “laboratory practices and procedures, quality control data security’’ and other critical areas. Wockhardt didn’t respond to a request for comment.
While bad laboratory practices are usually at the root of the suspect data that inspectors are finding all over the world, in India there have also been high-profile cases of “overt and deliberate fraud,” bringing the manufacturing culture there under particular scrutiny, said Janet Woodcock, the head of the FDA’s center that reviews drugs. Woodcock didn’t identify specific companies.
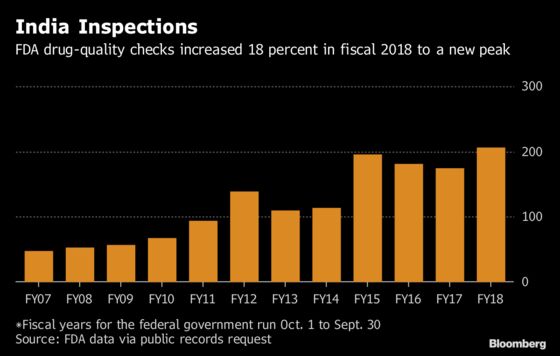
The agency increased inspections in India by 18 percent last year after two years of declines, underscoring the fact that India may pose the FDA’s biggest challenge as it struggles to ensure drugs are cheaper but also safe.
“I don’t think everything’s fine, and I’m upset about it, but there’s not much you can do,” because India’s low production costs makes its pharma industry “enormously important” to the U.S., said Bruce Downey, former chairman of an industry lobbying group who now sits on the board of Momenta Pharmaceuticals Inc., the Cambridge, Massachusetts-based generic drugmaker. “In New Jersey, there’s dozens of FDA inspectors. In India, there’s a dozen for the entire country.”
In a statement about the adverse-event reports, Mylan lawyer Mitchell J. Langberg said that “any conclusions, inferences or extrapolations about the quality of products made by Mylan based on this data is unreasonable and unreliable.” Mylan cited a disclaimer on the FDA’s website saying the number of such reports associated with a drug should not be used to determine the likelihood of a side effect occurring, and said the quality-control issues the FDA found at its factory in India did not impact the quality of its drugs.
The FDA uses the adverse-event-reports database to look for new safety concerns and monitor products after they’ve been approved. The reports have not been medically verified, according to the FDA.
What Bloomberg found in the agency’s records highlights what’s at stake in the questions that persist around industry-supplied data used to confirm the safety of generic drugs: When a doctor prescribes one, how do patients know it will work the way it should? If the data submitted by the drug’s manufacturer can’t be trusted, they don’t know.
Doubts about generic-drug companies’ data aren’t unique to India. But when you ask veterans of India’s pharmaceutical industry why the questions linger here, they use a singular term to explain it: jugaad.
The Hindi word, translated as “creative improvisation” when it was held up in the Harvard Business Review in 2010 as an example for corporate America, has been elevated to something of a national ideal in India. It’s credited with the rise of the country’s two global industries, technology and drugmaking. In both, Indian companies with far fewer resources than foreign rivals came up with cheaper, more effective ways of doing things that ended up being a competitive advantage.
But the word can have a darker connotation: Get it done at all costs.
“It’s kind of bending the rules, breaking the rules, and finding shortcuts—and in some cases, outright misdemeanors,” said Jagdish Dore, who runs pharmaceutical-industry consultancy Sidvim LifeSciences in Mumbai. “And the pharma industry is not one where you can bypass rules and regulations.”
People who closely follow the Indian business world trace jugaad’s emergence to the colonial era, when laws and regulations were often in service of British interests, rather than Indian ones, and so fair game to be skirted, bent or broken. After independence in 1947, the period of heavy handed government control and regulation of the economy known as the “License Raj” further cemented a flexible approach to the rules as a precondition for success in India.
In some ways, the country emerged out of a kind of state-sanctioned jugaad. Frustrated by the high price of drugs made by foreign firms, the Indian government tweaked its laws in the 1970s so that instead of a drug itself being protected, a company could patent only the process for making it.
That meant open season on any major brand-name drugmakers’ products, so long as the legions of highly trained scientists being pumped out of India's research institutes could find a different method for synthesizing them. By the time India opted back into the international patent system in 2005, the country had developed so much capacity for cheaply producing copycat drugs, it was poised to take over the global generics industry.
Today, India’s thousands of drug companies not only supply the country’s poor with affordable medicine, but have become a key source of drugs for the rest of the developing world. They played a central role in bringing down the price of lifesaving antiretroviral drugs that have helped contain the AIDS epidemic. Mylan’s plant is a supplier of these, too. Globally, India accounts for 20 percent of generic-drug exports.

But its drug industry also was responsible for one of the most dramatic examples of data manipulation in the history of the generics business. In 2013, New Delhi-based Ranbaxy Laboratories Ltd. pleaded guilty to the manufacture and distribution of adulterated drugs, and paid a $500 million penalty to the U.S. Justice Department, after widespread data manipulation was found at two of its factories.
India became a focus for the FDA, and regulators have uncovered a steady stream of suspicious behavior at numerous companies, from computer files found deleted to employees caught on a company’s own security cameras shredding documents the night before an inspection, according to FDA inspection documents posted to the agency’s website. The FDA calls these data-integrity problems.
The concerns got bad enough that Margaret Hamburg, who ran the agency under President Barack Obama, traveled to India herself in 2014 to get a handle on the problem. That year, the FDA stepped up oversight, resulting in additional sanctions in 2015, when Indian plants accounted for 60 percent of the 20 warning letters handed down from the FDA’s Office of Manufacturing Quality.
The regulatory scrutiny has taken a toll on the companies. FDA warning letters bar new product launches from a facility, restraining revenue growth. Since 2015, an index of Indian pharma company stocks has declined 25 percent on the Bombay Stock Exchange. The broader market, India’s benchmark S&P BSE Sensex Index, has rallied almost as much the other way over the same period.
That’s helped put pressure on the country’s pharma barons—a coterie of homegrown billionaires whose net worth is tied up to a large extent in their company's stock. In the last two years, the most important executives have gathered for annual forums at an upscale hotel in Mumbai to hold forth about the primacy of a “culture of quality” to an audience of enthusiastic middle managers and visiting FDA officials.
D.G. Shah, a white-haired veteran of Pfizer’s India operations who now runs the Indian Pharmaceutical Alliance, an industry body representing the largest domestic companies, has organized the panels. Shah said the root of the problem in India’s generics industry is cultural, but insisted a lax attitude toward process and procedure is to blame.
“That’s the lifestyle, that’s the way people live in this country,” he said in an interview in Mumbai. He takes as an example the perpetual traffic jam honking away outside his window, where motorists habitually violate the rules of the road, and high concrete barriers are in place to keep cars, motorcycles and scooters from seeking a shortcut by darting into oncoming traffic or up on the sidewalk.
“Culture change is not something that can happen overnight,” said Shah, whose group is undertaking training programs with its member companies to help a new attitude trickle down to all employees. “Culture and behavior change requires continuous effort.”
While the number of FDA inspections in India and China has fluctuated in recent years, India’s share of FDA warning letters from the agency's Office of Manufacturing Quality began to decline in 2016 as companies in China received more. Last year, out of 53 warning letters issued worldwide, 10 went to drug factories in India and 16 in China.
Shah said such figures are evidence that Indian companies have made progress ensuring that a disregard for the rules doesn’t make it to the factory floor.

In 2016, an FDA inspector visited Mylan’s flagship factory about an hour’s drive outside the ancient holy city of Nashik in western India, where it makes its version of Lipitor for the U.S. According to an agency warning letter, the inspector found that quality-control technicians there had disregarded about 72 percent of failing quality checks in a six-month period—for no good scientific reason. He couldn’t rule out the possibility that staff were retesting failing drugs until they passed.
Interviews with nine current and former employees at the plant produced more innocuous explanations for what contributed to the problem. They said investigating why a drug fails testing is time-consuming, and whether due to time pressure, ignorance or just laziness, it was easier to say the tester spilled some of the sample or made some other mistake and then test again. If the batch passed the second test, it was assumed to be fine.
The Mylan employees, who requested anonymity because they still work for the company or elsewhere in the pharmaceutical industry, said that members of the quality-control team at that plant faced constant pressure to get the drugs out the door as fast as possible. This made it harder to properly investigate why a batch failed.
Other times, technicians could be diligent about investigating the reason for a failed test, but not as thorough about filling out the report to document their work, either because they didn't know how or had difficulty with English. Some of the employees Bloomberg spoke to said plant managers had been aware of the problem and were instituting training courses to correct it at the time of the 2016 inspection.
Asked about the workers’ comments, Mylan spokeswoman Lauren Kashtan said all medications at the company’s facilities are made “under stringent processes, procedures and rigorous testing designed to ensure that they meet the highest standards for quality, safety and efficacy. Any explicit or implicit suggestion that Mylan employees circumvented data and quality systems that jeopardized the quality of the medications we manufacture—for time pressures or any other reason—is simply false.”

When FDA inspectors visited Mylan’s plant again last year, they made note of four potential violations of manufacturing guidelines, but the issue around invalidated test results was not one of them. One of the potential violations was a lack of soap and hand-drying equipment in the bathroom.
In the earlier statement from its lawyer, Mylan said last year’s inspection resulted in the lifting of the FDA warning letter against its plant issued after the 2016 audit. In the intervening time, the company spent “millions of dollars” on employee training and to have a third party audit and certify its investigation results, it said. Mylan said that it had not experienced serious quality-control issues with respect to the production of its generic version of Lipitor in India, and that it also manufactures that drug in the U.S.
Another FDA inspection of the Nashik plant about a week ago “found no repeat observations from prior inspections,” Kashtan said, and the facility was in compliance.
As for Mylan’s version of Lipitor being more likely to be associated with adverse events than other top-selling generic versions, the company said it’s more appropriate to compare its safety record with the original branded drug made by Pfizer Inc. Adjusted for the number of patients who use each version, Pfizer's is associated with adverse-event reports at a rate “thousands of times” more than Mylan’s, Mylan said.
When asked about the greater number of adverse-event reports for Pfizer versus makers of generic Lipitor, an FDA spokeswoman said it was because patients often don’t know they’re on a generic and identify their medicine by the well-known brand name of the company that invented it, rather than the drug’s chemical name used by generic companies who copy it. That means versions made by generic makers are often misidentified in the database, she said.
A spokesman for Pfizer, Thomas Biegi, said Lipitor, or atorvastatin calcium, “is the most-studied statin and has a well-established efficacy and safety profile. Since its approval in the U.S. more than 20 years ago, Lipitor has been prescribed to millions of patients and studied in more than 400 clinical trials.”
To help Indian factories change their culture in the wake of the Ranbaxy scandal, FDA leadership originally planned to put more inspectors on the ground. This proved harder than expected. Instead, the number of India staff shrank from a high of 12, in 2013. to only four at one point in 2017. Eight FDA employees are stationed at the India outpost now, including two drug inspectors, the agency said.
The original argument for having foreign offices was that staff would have a chance to learn the customs of the country and build key relationships with government officials to instill best practices, said the FDA’s Woodcock. Inspectors based in-country also don’t need a visa for each round of inspections, while those based in the U.S. do. Countries can use visas as a way to punish the U.S. for actions they don’t like, such as trade wars, and to protect their domestic businesses.
The problem is that the FDA can’t find people who want to live in India, Woodcock said. Difficulty hiring forced the agency to close its Mumbai office in 2016.
Because of those challenges, the agency has largely ditched plans to build up offices in India and China and is instead relying on a U.S.-based cadre of inspectors who fly overseas for a few weeks at a time, she said. This cadre was supposed to number as high as 20 people, but 12 were serving in its ranks as of December. The agency expects that number to go up to 17, and says any of its 255 drug inspectors in the U.S. can fly to foreign plants as well if needed.
Amid those staffing challenges, the number of so-called surveillance inspections the FDA has conducted in India, like the ones at Mylan’s plant, have fluctuated. The number fell 7 percent in fiscal 2016 and then a further 4 percent in fiscal 2017, to 174. But in fiscal 2018, it jumped 18 percent, to 206.
About 700 Indian factories are licensed for exporting to the U.S. They sprawl across a country that could fit Texas four and a half times, where 22 major languages are spoken. Getting tens of thousands of workers, spread from tiny villages to sprawling megacities, to change their habits is a herculean task.
Rishikesha Krishnan, a professor at the Indian Institute of Management in Bangalore, related a story of one foreign regulator visiting a drug factory of a company he declined to name. The inspector’s first question when he arrived after negotiating India’s chaotic roads was, “Guys, how can you maintain quality in your plant if this is the way things are outside?’”
“I think this is one of the challenges the pharmaceutical industry has always faced,” Krishnan said. “It’s very difficult to separate or segregate a particular industry or a particular factory from the society in which we live.”
—With assistance from Ed Strong, Rahul Satija, and Naomi Kresge
To contact the editor responsible for this story: Flynn McRoberts at fmcroberts1@bloomberg.net, Timothy Annett
©2019 Bloomberg L.P.