ChemoCentryx Up Most Since 2019 on Autoimmune Drug Approval
ChemoCentryx Soars Most Since 2019 on Autoimmune Drug Approval
(Bloomberg) -- ChemoCentryx Inc. shares surged the most since November 2019 after the drug developer won U.S. approval for Tavneos as a treatment for a rare autoimmune disorder.
The stock soared 96% on trading volume jumped to about 144 million shares, up from 6 million shares on Thursday. It’s added about $1.31 billion to a market value that now sits around $2.68 billion.
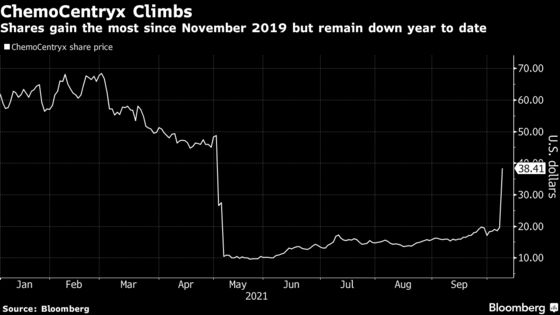
Even with Friday’s rally, the stock is still down about 38% this year. ChemoCentryx tumbled by a record 62% in May after receiving narrow backing from an FDA advisory panel in support of the benefit-risk profile for its avacopan drug candidate.
The company expects a regulatory decision regarding Tavneos in Europe by the end of the year following the European Medicines Agency review. The drug, for ANCA-associated vasculitis, won approval in Japan late last month.
The drug approval prompted Piper Sandler to upgrade ChemoCentryx to overweight from neutral, with analyst Edward Tenthoff describing the news as a “positive surprise.” Raymond James analyst Steven Seedhouse also raised ChemoCentryx to strong buy from outperform and lifted his price target on the stock to a Street-high $107 from $62. The stock has seven buy ratings, one hold and one sell, and an average 12-month price target of $58, according to data compiled by Bloomberg.
©2021 Bloomberg L.P.