Solid Bio Plunges on Mounting Gene Therapy Safety Issues
Solid Bio Plunges on Mounting Gene Therapy Safety Issues
(Bloomberg) -- Shares of Solid Biosciences Inc fell as much as 41% Tuesday to a record low after the company reported serious side effects for a patient receiving its gene therapy treatment, SGT-001, for Duchenne muscular dystrophy. Two analysts reacted by lowering their investment ratings to sell.
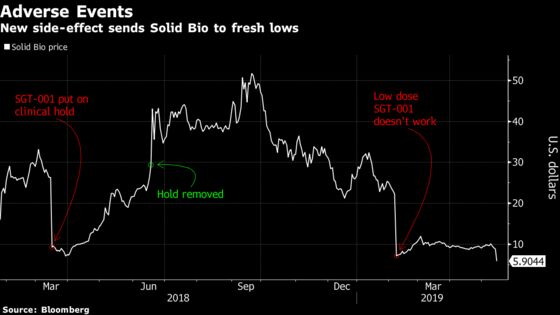
With Sarepta Therapeutics Inc and Pfizer Inc working on competing treatments for Duchenne muscular dystrophy, Solid’s SGT-001 is “competitively suboptimal,” Goldman Sachs analyst Salveen Richter wrote in a note cutting Solid Bio to sell from neutral and lowering the 12-month price target by $1 to $4.
Goldman had previously “expressed concern” over the higher SGT-001 dose, “which now appears to have been confirmed”. SLDB’s cash position is also a concern, Richter said.
Credit Suisse’s Martin Auster cut Solid to underperform from neutral, and lowered his price target to $6 from $7, writing “enrollment at this high dose may prove challenging.” While improved efficacy at the higher dose could offset the gene therapy's side effects, ``promising initial data from Sarepta and positive commentary from Pfizer” will limit Solid’s upside.
One analyst tracked by Bloomberg still rates Solid a buy, while four rate it a hold, and three rate it a sell. The average price target is $9.50.
To contact the reporter on this story: Cristin Flanagan in New York at cflanagan1@bloomberg.net
To contact the editors responsible for this story: Catherine Larkin at clarkin4@bloomberg.net, Steven Fromm
©2019 Bloomberg L.P.